J Pharm Pharmaceut Sci (www.ualberta.ca/~csps) 2(3):99-107, 1999
A Novel Skin Penetration Enhancer: Evaluation By Membrane Diffusion And Confocal Microscopy
Received manuscript October 8th 1999, Revised November 29th, 1999; Accepted November 30th, 1999.
Saunders
James CJ1,
Davis Henry J, Coetzee Linda, Botha Susan, Kruger Ansie E,
Meyer
Zall Laboratories, Drie Valleyen, Sedgefield, South Africa
Grobler
Anne,
Medical
Research Council, Cape Town, South Africa
PDF Version for printing
Abstract
Purpose. The aim of this study was to determine the in vitro transdermal efficacy of a Meyer Zall Laboratories (MZL) oil/water emulsion in two separate preparations containing the actives, coal tar and the non-steroidal anti inflammatory drug, diclofenac sodium. Method: The release rate of the two active ingredients from MZL dermatological preparations, Exorex and Athru-Derm and four comparator products was determined using an enhancer cell system, whilst specific penetration characteristics of the MZL formulation were elucidated using confocal and electron microscopy. The latter properties were explored at both the organ level, using human skin, as well as at a cellular level using a melanoma cell line. Results: While the in vitro release rates for all formulations was high, coal tar and diclofenac release from Exorex and Athru-Derm respectively was, at nearly all time intervals, significantly higher than from comparator products. Microscopy revealed the presence of spherical liposomal type structures in both the MZL lotion and a comparator gel. In the MZL lotion, the majority of these structures, referred hereto as emzaloid particles, were in the order of magnitude of about 50 nm to 1 µm in diameter with a small minority exceeding these dimensions. After application of Athru-Derm to human skin, intact emzaloid particles of submicron dimensions were detected in the epidermis in association with the cell membranes. The affinity of the MZL lotion for cell membranes was further demonstrated with melanoma cells; in addition, the formulation was seen to penetrate even to the nucleus of viable cells. Conclusion: Overall the data suggest that the oil/water base in MZL formulations is a highly efficient transdermal vehicle able to transport a wide range of indication- specific actives to their site of action.
Introduction
The scope of this study was to evaluate the efficacy of the Meyer Zall (MZL) base formulation by comparing the in vitro release rates of two known actives, coal tar and diclofenac from several market competitors including MZL Exorex lotion and topical antiinflammatory, Athru-Derm lotion (1% with respect to diclofenac) and to explore possible mechanisms of penetration using confocal laser scanning microscopy (CLSM). The MZL base formulation refers to a transdermal drug delivery system developed at Meyer Zall Laboratories, trademarked under the name, Emzaloid delivery system." This formulation is used in several preparations for the treatment of many skin disorders such as psoriasis, eczema, and dermatitis, and contains ethyl esters of the essential fatty acids, linoleic acid and linolenic acid, as well as oleic acid, emulsified in water saturated with nitrous oxide. Based on empirical clinical observations, it was proposed that the MZL base formulation not only maintains the membrane integrity of cells of the epidermis, but is also a highly effective transdermal carrier of indication specific drugs. In order to test this, a membrane diffusion study was carried out. Such studies, utilising enhancer cell types, are routinely used to test relative efficacies of transdermal delivery systems (1, 2). Later, in an attempt to elucidate the actual underlying mechanism and route of transport, CLSM was used to a) determine structural components in MZL formulations and to compare these with a leading brand of topical application, and b) visualise the interaction of the formulation with human skin samples and a human melanoma cell line. CLSM is a powerful technique that permits direct visualisation of penetration pathways in unfixed skin samples. Up to now, the interaction of liposomes with skin has been well documented using both freeze fracture electron microscopy (3, 4) and CLSM (4, 5, 6). The technique has also been used to compare penetration depths of a fluorophore into different skin preparations (7). Human melanoma cells were used in this study to elucidate possible mechanisms of interaction of the formulation at a cellular level. The use of a laser as an energy source enables the microscope to be used as an optical knife that can optically section a sample at varying depths; in addition, dynamic changes can also be visualised in real time. This is achieved by labeling the sample with a fluorescent marker, a molecule that enters an excited state during laser interaction and emits light of a specific wavelength.
Materials And Methods
Materials: The in vitro release from the lotion (gel) preparations was measured with a 57-6M Hanson Model diffusion cell test system (Hanson Research Corporation: Chatsworth, California, USA), related to the USP dissolution apparatus described elsewhere (8). The diffusion cell assembly includes a donor top and receptor chamber. The donor top consists of a drug dosage wafer, an acrylic top plate, and a clamp to connect top to bottom Abdominal skin grafts were obtained from Panorama hospital (Cape Town, South Africa); human melanoma cells (UCT-Mel1: Cape Town, South Africa), the characterisation of which is described elsewhere (9); pen/strep (GIBCOBRL, Life Technologies: Paisley, Scotland) prepared by adding 5000 units of penicillin G with 5000µg/ml streptomycin sulphate in normal saline; foetal calf serum 10% (GIBCOBRL, Life Technologies: Paisley, Scotland), obtained from raw bovine samples which were both virus and mycoplasma tested, and EC approved culture medium, DMEM HAMS F12 (GIBCOBRL, Life Technologies: Paisley, Scotland) were utilised in the cell culture study. The fluorophores, Nile Red, Alexa- 568 and TMRM (tetramethylrhodamine methyl) were purchased from Molecular Probes (Leiden, Holland). The two MZL preparations used were Athru-Derm (containing 1g/100g diclofenac sodium), and Exorex (containing 1g/100g coal tar). These are both lotion preparations consisting of an oil/water emulsion base (Meyer Zall Laboratories, Sedgefield, South Africa). Athru-Derm was compared with a competitor topical anti-inflammatory gel, Voltaren Emulgel (Novartis; obtained locally, Johannesburg, South Africa). Voltaren Emulgel contains diclofenac diethylammonium equivalent to 1g/100g diclofenac sodium. Exorex lotion was compared with Clinitar (1% coal tar: Shire Pharmaceuticals, Andover, England), Polytar (10% coal tar: Stiefel Laboratories, Bucks, England) and Gelcotar ( 10% coal tar: Quinoderm Ltd, Oldham, England), all competitor products used in psoriasis treatment.
Membrane Diffusion: The release experiment was carried out six fold for each preparation and the average release was calculated for each release point. The reservoir of the dissolution cell (enhancer cell) was filled with the lotion and covered with the membrane (pig skin excised from the jawbone), taking care to exclude air bubbles between the gel and the membrane. The cell was capped and placed in the dissolution vessel containing the receptor medium (pH 6.8 phosphate buffer; 12 ml). The receptor medium was maintained at a constant temperature of 32ºC. The paddle speed was 100 rpm. Samples of 250 µl were withdrawn with a micropipette at different time intervals up to a maximum of 90 minutes. The samples were analysed for diclofenac by means of HPLC, for coal tar, by means of UV. The specificity, accuracy, and precision of these methods were validated in accordance with the USP (10 ).
Calculations: Cumulative concentration of actives released was calculated as: Concentration of active released + surface area of exposed skin. The average release plus minus the standard deviation was expressed for each time point, and displayed graphically, except in the case of Gelcotar and Polytar. The release rate was calculated as: Accumulative dose (µg released/ cm²) + time. The release rates were calculated for each time point, and expressed as an average of all time points for each product. Significance was determined using Students t-test. P values <0.05 were considered to be significant.
Preparation of samples for microscopy: Human abdominal skin grafts (50 200 µm thick) were cut using a dermatome and immediately placed in DMEM HAMS F12 medium. Samples were then kept refrigerated until experimented upon, this occurring within a few hours of the operation. Small sections (roughly half a cm²) were then cut in a sterile environment. Human melanoma cells were cultured directly on coverslips used for microscopy.
Microscopy: The confocal microscope system used was a Zeiss 410 LSM model equipped with a Krypton/Argon laser (objectives: Plan Achromat. oil immersion; numerical aperture of 1.4). The fluorophone, Nile red was used in the initial formulation and skin preparations. In these studies, Nile red was chosen as the fluorophone as it predominantly accumulates in lipid rich domains, and therefore readily stains phospholipid components of the formulations. A stock solution of 200 mM Nile red in dimethyl sulphoxide (DMSO) was thoroughly mixed with the Athru-Derm and Voltaren Emulgel to produce a final working solution containing 10 mM Nile red. In the comparative study of formulations, sample droplets were placed directly onto a 15 µm thick coverslip which was then fixed into a sample chamber and subjected to CLSM. Since conclusions based on CLSM technology are restricted to the fluorescent dye in question, cryo-electron microscopy was then used to confirm findings made with the CLSM. In these experiments, samples were frozen in liquid nitrogen at 90ºC and then fractured. After sublimation of the ice lattice surface, remaining structures embedded in the surface could then be visualised directly. To visualise the interaction of formulation with skin, skin samples were placed into the Athru-Derm lotion containing Nile red. After five minutes samples were then gently wiped clean with a tissue and mounted on the coverslip for observation in the CLSM system. Nile red penetration in the presence of Athru-Derm formulation was then compared with a control solution containing 10 mM Nile red in DMEM culture medium. In this study, the comparatives were applied topically on skin samples and left for a period of ten minutes before visualising the respective penetration depths of Nile red. In the melanoma cell preparation, two separate molecular probes were used in an attempt to visualise the ability of the base MZL formulation in Athru-Derm to deliver diclofenac to its site of action on the cell membrane. The stain specific for cell membrane potential, TMRM (25 µM in DMEM HAMS F12), was used to confirm the viability of cells. The cells were stained in the TMRM for 30 minutes. An Alexa-568diclofenac conjugate was then formed by adding 10 µl of a 50 mM Alexa stock solution to 90 µl of a 50 mM diclofenac stock solution, ensuring that enough excess diclofenac was present to bind with the Alexa. The conjugate was incubated for an hour at 37ºC, before being added to the base formulation. To implement an effective control, the interaction of the Alexa-diclofenac conjugate with the cell membranes was compared before and after the addition of MZL base formulation. In both cases, the final concentration of Alexa 568 in the preparation was 25 µM.
Results
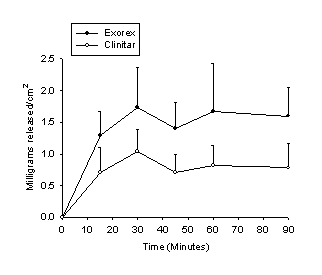
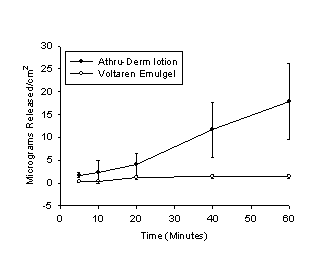
Membrane diffusion: Significantly more coal tar is released at each time interval from Exorex than Clinitar (fig. 1). The average release rate of coal tar from Exorex was 2.64 mg/cm²/h ± 1.66, and from Clinitar, 1.44 mg/cm²/h ±.977. The data also indicate that significantly more diclofenac is released from Athru-Derm than Voltaren Emulgel (fig. 2), at all time intervals except for the ten minute interval (n=5). The average release rates were 33.11 µg/cm²/h ± 4.78 from Athru-Derm, and 4.99 µg/cm²/h ± 2.04 from Voltaren Emulgel.
Fig. 1: Cumulative release of coal tar from two different preparations. The error bars represent the standard deviation of the mean. Coal tar release from Exorex was significantly greater at all time points.
Fig. 2 Cumulative release of diclofenac from Athru-Derm, and comparator, Voltaren Emulgel. The error bars represent the standard deviation from the mean. Release of diclofenac from Athru-Derm was significantly greater at all time points except for the ten minute point.
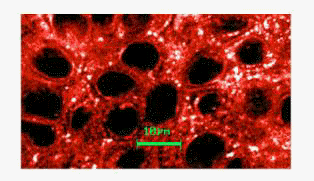
Fig.3 a) Emzaloid particles observed in Athru-Derm using CLSM (magn.=63, zoom×2.5;excitation =568 nm; long pass filter: 590 nm). Note the range of particle sizes (about 50 nm 5 µm) and striking contrast with the background, demonstrating high affinity with the lipophilic Nile red. The spherical structures observed in Voltaren Emulgel (b) are nearly uniform in size and considerably larger than emzaloids.
Human skin: CLSM data revealed spherical microstructures in both Athru-Derm and Voltaren Emulgel (fig.3). However a marked difference in size and distribution was noted for the two formulations. In Athru-Derm the structures (now referred to as emzaloid particles) were smaller in diameter, the majority measuring between 50nm and 1 µm. A small minority was over a micron in diameter, and some of even up to 5 µm were visualised. Emzaloid particles also associated readily with the Nile red as demonstrated by their marked contrast with the background. Cryo-electron micrographs (fig.4) confirmed the observations made in fig.3. Further variations in the electron microscopy technique revealed that the molecular density of these structures was different to the surrounding frozen lattice.
Fig.4 Cryo-electron microscopic confirmation of particle size difference in Athru-Derm (a), and Voltaren Emulgel (b).
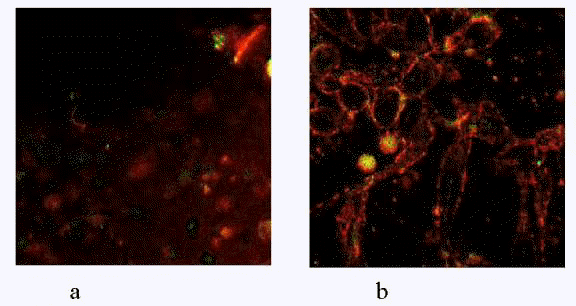
An optical section a few microns in depth through the epidermis after exposure to the Athru-Derm-Nile red conjugate (fig.5) revealed the presence of intact emzaloid particles associated with the membranes, which could be seen to move laterally in time. Furthermore only those emzaloid particles smaller than a micron in diameter could be observed in these regions. In the depth study, the Nile red associated with Athru-Derm was visualised at a skin depth at least 4 times that of the Nile red in the control (fig.6).
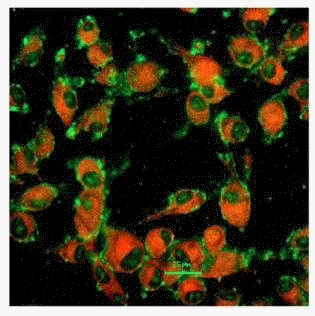
Human melanoma cells: The Alexa 568 diclofenac conjugate in the presence of Meyer Zall formulation has a strong affinity for the cell membranes of the melanoma cells, as illustrated by the green fluorescence (fig.7).
Fig.5 Image obtained by optical sectioning within the epidermis after the skin was soaked in the Nile red dyed Athru-Derm (magn.=63, zoom×2). Note the presence of submicron emzaloids in the extracellular regions associated with the membranes. The cell nuclei (dark spaces) did not take up Nile red at all. Larger emzaloids (> 1 µm) were not present in this optical plane.
The
normally cell-impermeant Alexa stain is not only localised to specific
regions on the membranes, but also appears to have penetrated the
nucleus. The TMRM stain (shown as red) indicates the cells have an
appreciable membrane potential and are therefore viable. In a control
study with and without formulation (fig. 8), there appeared to be little
or no binding with the cell membranes in the absence of formulation.
After addition of formulation, the Alexa-diclofenac conjugate was
readily taken up by the cell membranes.
Discussion
Although the release rate of coal tar for most formulations tested was high (fig.1 for example) , the data presented in this paper suggest that the MZL preparations, Exorex and Athru-Derm have a significantly greater efficacy in the transdermal delivery of coal tar and for the most part, diclofenac than comparator products, in pig skin. In preliminary observations of membrane diffusion results obtained from Gelcotar and Polytar, it was of interest to note that release of coal tar from these 10% preparations was in the same order as Exorex, in spite of higher concentrations of the active ingredient (unpublished preliminary data).
Fig.6 Comparison of penetration depths of the Nile red control (a) and MZL base formulation preparation (b) measured in the skin ten minutes after topical application (magn=63×2). Note how the Nile red penetrates at least four times deeper (33 µm) in the presence of MZL base formulation, when compared with the control (9 µm). In b) the surface layer of the skin could not be visualised because the laser intensity was such that the Nile red fluorescence was too high to identify specific structures. The first frame at 0µm therefore shows nucleated cells uncharateristic of stratum corneum, and probably corresponds to a more accurate depth of about 10 µm.
Fig. 7. Human melanoma cells stained with TMRM (red) and Alexa- 568 (green) in the presence of Athru-Derm (magn=63; exc.=568 nm; long pass filter: 590 nm). The Alexa- 568 is bound to diclofenac and appears to associate with specific localised regions on the cell membrane. The normally cell-impermeant Alexa-568 is also seen within the cell nucleus.
It has been proposed that fluidisation of intercellular domains and thus a structural modification of the stratum corneum is a possible mechanism for the enhanced transport of encapsulated drugs in vesicles (11). With regard to liposomes used in dermatological preparations, a number of physico chemical properties have been defined as being important determinants in the effectiveness of transdermal penetration. These properties include the size of vesicles, surface charge, fluidity (often referred to as elasticity), and the heterogenous structure of the liposome bilayer (12, 13, 14). It is proposed that these features should enable liposomes to squeeze themselves through intercellular regions of the stratum corneum under the influence of the transepidermal water-activity gradient. In the present study, a significant observation is, that although particles ranged in size from about 50 nm to 5 µm in the Athru-Derm, only the smaller particles of submicron dimensions could be visualised within the epidermis. Since particle size is an important criterion for transdermal penetration, the fact that larger microspheres could not be visualised in the epidermis may be a possible explanation why the release rate of Voltaren Emulgel (consisting of larger particles) was, for the most part, significantly less than that of Athru-Derm (see fig. 3).
Fig. 8. Human melanoma cells stained with Alexa-568 in the absence (a) and the presence (b) of formulation. The Alexa dye appears in this case as a red stain. In the control (a) the stain does not associate with the membranes, appearing as a diffuse red. The Alexa/emzaloid conjugate, however, is quickly and readily taken up by the cell membranes (b).
After the membrane diffusion studies were performed, it was hypothesised that the enhanced transdermal transport rate of MZL formulations was the result of an increase in the fluidity of the membranes and intercellular lipids, caused by the anaesthestic action of nitrous oxide in the formulation (15), and that transport, therefore, occurs in close association with the membranes. As revealed by CLSM, emzaloid particles could not only be visualised in association with the cell membranes (fig. 5), but could also be visualised migrating within the epidermis in real time. These observations are consistent with the above-mentioned hypothesis. Indeed the presence of nitrous oxide in the formulation may enhance both intercellular lipid fluidity, as well as the fluidity of the emzaloid particles. Both of these are important determinants of efficient transdermal penetration. It follows then that emzaloid particles may be able squeeze themselves through intercellular regions of the stratum corneum as intact structures, and by this mechanism, deliver encapsulated drugs to the dermis. Another possible mechanism was postulated in a similar study using freeze fracture electron microscopy to observe the interaction of liposomes and niosomes with human skin where both large and small vesicular structures were observed in the stratum corneum (3). In this case, it was suggested that single molecules which make up such structures might penetrate the intercellular spaces in dispersed form, and at certain regions in the stratum corneum, are able to accumulate and reform into microspheres. Although emzaloid particles can be considered to fall into the same general category as liposomes, niosomes or submicron emulsions, they differ both in constitution and preparation techniques (3, 16, 17) and are thus regarded as unique. Results in the present study indicate that diclofenac in the presence of the MZL formulation readily associates with cell membranes in cell culture preparations (fig. 8). It is probable therefore that the diclofenac is associated with emzaloids in Athru-Derm. Since the action of diclofenac is at the level of the cell membrane (18), the localised concentrations of Alexa stain observed in fig.7 may correspond to receptor rich regions on the membrane. It is of interest to note, that the normally cell-impermeant Alexa dye is able to penetrate the cell membrane and stain the nucleus when it is administered with formulation. For years, liposomal drug delivery systems have been developed to solve problems of drug solubility, instability and rapid degradation (19). The emzaloid delivery system could also conceivably find application as a delivery system of a wide range of drugs for parenteral administration, including those whose primary action is in the nucleus, or as non-viral vectors for gene therapy.
Properties of good skin penetration enhancers are well described (20). Amongst others they should be pharmacologically inert, non-toxic, non-irritating, and should be compatible with a wide range of ingredients in various dosage forms. The MZL formulation used in Athru-Derm fits these criteria. Moreover, the emzaloids and associated essential fatty acids in the formulation likely play a role in replenishing a lipid deficiency in the skin by incorporating phospholipids, maintaining the integrity of the epidermal permeability barrier (3, 21, 22). The oleic acid in the MZL formulation may also play a role in enhancing transdermal penetration by temporarily disrupting the packed structure of the intercellular lipids because of the incorporation of its kinked structure (20).
The unique penetrative properties of MZL emzaloid technology may offer further attractive applications in pharmacotherapy and cosmetics; Not only can a faster onset of drug action be achieved, using a lower concentration of drug in the formulation, but topical application has additional safety value, rejuvenating skin lipids without the irritation characteristic of many other transdermal penetration enhancers. It is furthermore reasonable to speculate that the apparent ability of emzaloids to penetrate tissue as intact structures might overcome a major limitation of liposomal technology, that being the extravasation of liposomes from the vascular compartment.
Acknowledgements
We are deeply indebted to: Piet Meyer, chairman of Meyer Zall Laboratories, and Steven Zall for their continued support and interest in the undertaken study; The South African Medical Research Council for generously offering their laboratory facilities and related expertise. The human skin grafts were kindly donated by Dr. Luckhoff of the Panorama Hospital.
References
-
Ramis, J.; Conte, L.; Segado, X.; Forn, J.; Lauroba, J.; Calpena, A.; Escribano, E. and Domenech, J., Influence of formulation on the in vitro transdermal penetration of flutrimazole. Arzneimittelforschung, 47(10): 1139-44, 1997.
-
Said, A.; Makki, S.; Muret, P.; Humbert, P. and Millet, J., Psoralens percutaneous permeation across the human whole skin and the epidermis in respect to their polarity. J. Dermatol. Sci., 14(2): 136-44, 1997.
-
Junginger, HE.; Hofland, HEJ. and Bouwstra, JA., Liposomes and niosomes: Interactions with human skin. Cosmetics and Toiletries 106 (8): 45-46, 1991.
-
Zellmer, S.; Reissig, D. and Lasch, J., Reconstructed human skin as model for liposome-skin interaction. J. Controlled release. 55(2-3): 271-9, 1998.
-
van Kuijk-Meuwissen, ME.; Mougin, L.; Junginger, HE. and Bouwstra, JA., Application of vesicles to rat skin in vivo: a confocal laser scanning microscopy study. J. Controlled Release, 4;56(1-3): 189-196, 1998.
-
van Kuijk-Meuwissen, ME.; Junginger, HE. and Bouwstra, JA., Interactions between liposomes and human skin in vitro, a confocal laser scanning microscopy study. Biochim. Biophys. Acta.,1371 (1): 31-9, 1998.
-
Simonetti, O.; Hoogstrate, AJ.; Bialik, W.; Kempenaar, JA.; Schrijvers, AHGJ.; Bodde, HE. and Ponec, M., Visualisation of diffusion pathways across the stratum corneum of native and in-vitro-reconstructed epidermis by confocal laser scanning microscopy. Archives of Dermatological Research, 287: 465-473, 1995.
-
Fares, HM. and Zats, JL., Measurement of drug release from topical gels using two types of apparatus. Pharmaceutical Technology, 19: 52-58, 1995.
-
Hoal-van Helden, EG.; Wolson EL. and Dowdel EB., Characterisation of seven human melanoma cell lines: Melanogenesis and secretion of plasminogen activators. British Journal of Cancer. 54: 287-295, 1986.
-
The United States Pharmacopeia (United States Pharmacopeial Convention), National Publishing, Philadelphia: 1225: 2149-2151, 1999.
-
Rougier, A.; Dupuis, D.; Lotte, C.; Roguet, R. and Schaefer, H., In vivo correlation between stratum corneum reservoir function and percutaneous absorption. J. Invest. Dermatol. 81: 275-278, 1983.
-
Vrhovnik, K.; Kristl, J.; Sentjurc, M. and Smid-Korbar, J., Influence of liposomal bilayer fluidity on the transport of encapsulated substance into the skin as evaluated by EPR. Pharm. Res. 15(4): 525-530, 1998.
-
Cevc, G.; Gebauer, D.; Stieber, J.; Schatzlein, A. and Blume, G., Ultraflexible vesicles, transfersomes, have an extremely low pore penetration resistance and transport therapeutic amounts of insulin across the intact mammalian skin. Biochim. Biophys. Acta. 1368(2): 201-215, 1998.
-
Reinl, HM.; Hartinger A.; Dettmar, P. and Bayerl, TM., Time-resolved infrared ATR measurements of liposome transport kinetics in human keratinocyte cultures and skin reveals a dependence on liposome size and phase state. J. Invest. Dermatol. 105: 291-295, 1995.
-
Devlin, TM. Textbook of Biochemistry with Clinical Correlations, Wiley Liss Publishing Company, New York: 213-234, 1993.
-
Benita, S. and Levy, MY., Submicron emulsions as colloidal drug carriers for intravenous administration: Comprehensive physicochemical characterisation. Journal of Pharmaceutical Sciences, 82 (11) 1069-1080, 1993.
-
Schwarz, JS.; Weisspapir, MR.; Shani, A. and Amselem, S., Enhanced antiinflammatory activity of diclofenac in Jojoba oil submicron emulsion cream. J. Applied Cosmetology, 14: 19-24, 1999.
-
Vane, JR. and Botting, RM., Mechanism of action of antiinflammatory drugs. Int. J. Tissue React. 20(1): 3-15, 1998.
-
Allen, TM., Liposomal drug formulations. Rationale for development and what we can expect in the future. Drugs, 56 (5): 747-756, 1988.
-
Touitou, E.; Junginger, HE.; Weiner, ND.; Nagai, T.; Mezei, M., Liposomes as carriers for topical and transdermal delivery. J. Pharm. Sci., 83 (9): 1189-203, 1994.
-
Elias, PM. Membranes, lipids and the epidermal permeability barrier, in: R. Marks and E. Christophers ( Eds.), The Epidermis in Disease, MTP press, Lancaster, pp. 1-30, .
-
Elias, PM. and Friend, DS., The permeability barrier in mammalian epidermis. J. Cell Biol., 65: 180-191, 1975.
Corresponding
author: James Saunders, Meyer Zall Laboratories, PO Box 1456
Drie Valleyen, Sedgefield 6573, jcjs@mzl.co.za
Keywords: Confocal laser scanning
microscopy; Membrane diffusion; Fluorophore;
Transdermal penetration; Liposomes.
Published by the Canadian Society for Pharmaceutical Sciences.
Copyright © 1998 by the Canadian Society for Pharmaceutical Sciences.
http://www.ualberta.ca/~csps