J Pharm Pharmaceut Sci (www.cspscanada.org) 9(2):190-197, 2006
Epinephrine Infiltration on Nasal Field Causes Significant Hemodyna- mic Changes: Hypotension Episode Monitored by Impedance-cardio- graphy under General Anesthesi
Jian-jun Yang1, Jun Zheng2, Hong-jun Liu1, Yu-xiu Liu2, Jin-Chun Shen1, Zhi-qiang Zhou1
1Department of Anesthesiology, Jinling Hospital, Nanjing University, Nanjing, P.R. China; 2Department of Scientific Research Management, Jinling Hospital, Nanjing University, Nanjing, P.R. China
Date received April 17 2006 Date Revised June 22 2006 Date accepted June 2006 Date published July 2 2006
Corresponding Author: Dr. Zhi-qiang Zhou,, Department of Anesthesiology, Jinling Hospital, People's Republic of China. Email zbfcj@hotmail.com
ABSTRACT: PURPOSE: Local infiltration of epinephrine-containing local anesthetics is widely used in clinics particularly in the procedure of surgeries on vascularity field to provide good analgesia and hemostasis. A prospective randomized double blind control study was designed to observe hemodynamic changes caused by local infiltration of epinephrine- containing lidocaine solution on nasal field under general anesthesia. METHODS:90 adult patients undergoing elective functional endoscopic sinus surgery under general anesthesia were randomly allocated into three groups and received 1% lidocaine 4 mL with different dose of epinephrine (group I 20 µg; group II 40 µg; and group Ⅲ 0 µg) respectively. Mean arterial pressure (MAP), heart rate (HR), cardiac index (CI), systemic vascular resistance index (SVRI), and acceleration index (ACI) were recorded through impedance-cardiography at every 45 seconds in 6minutes after the beginning of local infiltration. RESULTS:Compared with the intra-group baseline, statistically significant hemodynamic changes particularly decrease in MAP with increase in HR at 1.5 minutes time point (P<0.01), and decrease in SVRI and increase in CI, ACI at and from 1.5 minutes time point (P<0.05) were observed in group I and group II, but not in group III. CONCLUSION: Local infiltration of epinephrine-containing lidocaine solution on nasal field causes significant decrease in MAP and SVRI, and increase in HR, CI and ACI.
Introduction
Inclusion and Exclusion Criteria of Patient
90 adult patients undergoing elective FESS, between the age of 18 and 60 years were enrolled in this study. Our study was approved by the Hospital Ethics Committee and conducted according to the Helsinki Declaration and written informed consents were obtained from all the patients.
The patients were all ASA physical status I or II without premedication. Patients with a history of coronary artery disease, hypertension, arrhythmia, and diabetes were excluded from the study. Moreover, patients whose mean arterial pressure (MAP) fluctuation (maximal MAP-minimal MAP) was above 10 mmHg in 3 minutes before local infiltrations were excluded either. 90 patients were randomly allocated into three groups with 30 cases each and received 1% lidocaine 4 mL with different dose epinephrine: group I with 20 µg (5 µg/mL); group II with 40 µg (10 µg/mL); and group Ⅲ (controlled group) without epinephrine.
Anesthesia Techniques
Total intravenous anesthesia (TIVA) was used in our study with premedication of scopolamine 0.3 mg intramuscular injection 30-40 minutes before anesthesia in operation room. Anesthesia was induced with midazolam 0.02 mg/kg, target controlled infusion(TCI) with propofol 4 µg/mL and remifentanil 6 ng/mL. Rocuronium 0.8 mg/kg was used to facilitate orotracheal intubation. Anesthesia was maintained with propofol 4 µg/mL and remifentanil 4 ng/mL. Tramadol 2 mg/kg with ondansetron 8 mg was intravenously infused 20 minutes before the end of the operation. All the patients were mechanically ventilated to keep the end tidal carbon-dioxide tension around 30–35 mmHg.
Epinephrine Infiltration
An appointed anesthetist filled a prescription of the different solutions; neither the surgeons nor the other anesthetists including the observer knew which solution was used. 25 minutes after the induction of general anesthesia and endotracheal intubation, the surgeon decongested the nose with four cotton swabs containing oxymetazoline and lidocaine for 3-4 minutes, then applied the local infiltration on nasal septum,middle nasal meatus, and inferior nasal concha before surgery and all the injection was accomplished in 20-35 seconds using the same size of hypodermic needle. Plasma expanders (hetastarch 3-6 mL/kg) and crystalloid (Ringer’s lactate 3-6 mL/kg) were infused in 30 minutes after endotracheal intubation to maintain MAP fluctuation (maximal MAP-minimal MAP) below 10 mmHg in 3 minutes before infiltration.
Measurements of Hemodynamics
Hemodynamic values including MAP, heart rate (HR), cardiac index (CI), systemic vascular resistance index (SVRI), and acceleration index (ACI) were recorded at 45-second-interval in 6 minutes at the following nine time points by ICG (Bioz, CardioDynamics, THE ICG COMPANY, San Diego, California, USA): baseline, 0.75 minutes, 1.5 minutes, 2.25 minutes, 3 minutes, 3.75 minutes, 4.5 minutes, 5.25 minutes, and 6 minutes after the beginning of local infiltration.
Statistical Analysis
Statistical analysis was performed by statistics package for social science (SPSS) of 11.0-version. Quantitative data were expressed as mean ± SD and qualitative data were expressed as proportions. Patient characteristics and volume of intravenous fluid infusion before local infiltration were analyzed by Chi–square test or one-way analysis of variance (ANOVA).
After a test for homogeneity of related variances, inter-group comparisons were made using one-way analysis of covariance (ANCOVA) (dependent variable: hemodynamic value; fixed factor: group; covariate: the baseline of hemodynamic value) followed by Student-Newman-Keuls test for post-hoc multiple comparisons. Intra-group comparisons were made using one-way ANOVA for repeated measurements followed by Student-Newman-Keuls test for post-hoc multiple comparisons. Differences were considered to be significant at p<0.05.
RESULTS
Study Population
Six patients were excluded from the analysis. Two of them were due to the MAP fluctuation above 10 mmHg in 3 minutes before infiltration, and the other four were due to data loss at some time points because of spending too long time in waiting the MAP measurement. So 84 patients completed this study in three groups: group I (n=29); group II (n=27); and group Ⅲ (n=28). There was no significant difference between three groups with regard to age, sex, weight, body surface area (BSA), ASA physical status, and volume of intravenous fluid infusion before local infiltration. (Table 1).
Table 1. Patient characteristics and volume of intravenous fluid infusion before local infiltration. Data are expressed as mean ± SD or proportions. There was no significant difference between Group I, Group II and Group III using Chi–square test or one-way ANOVA. BSA= body surface area; ASA=American Society of Anesthesiologists.
|
Group I |
Group II |
Group III |
Age (yr) |
32±12 9.7±1.5 |
37±13 9.9±1.7 |
37±14 10.1±1.6 |
Hemodynamic Changes
MAP, HR, CI, SVRI, and ACI recorded at 45-second-interval in 6 minutes after the beginning of local infiltration at nine time points were expressed and analyzed. Compared with the intra-group baseline, significant hemodynamic changes were observed in group I and group II but not in group Ⅲ,including: decrease in MAP with increase in HR at 1.5 minutes time point (P<0.01); decrease in SVRI and increase in CI, ACI at and from 1.5 minutes time point (P<0.01 or P<0.05, the only exception was SVRI at 3 minutes time point in group II). Similar results also were observed in group I and group II compared with group III at the same time point between groups but ACI in group I.(Figure 1-Figure 5).
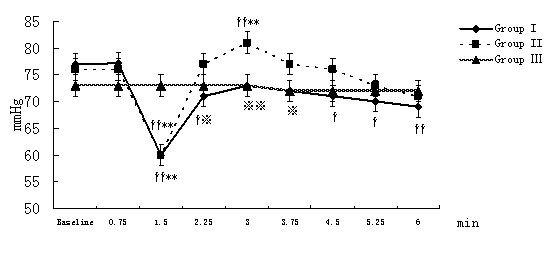
Figure 1. Changes in MAP at different time points after the beginning of local infiltration
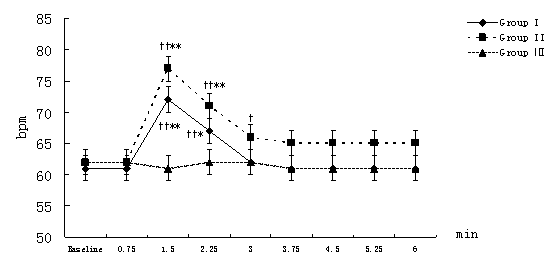
Figure 2. Changes in HR at different time points after the beginning of local infiltration
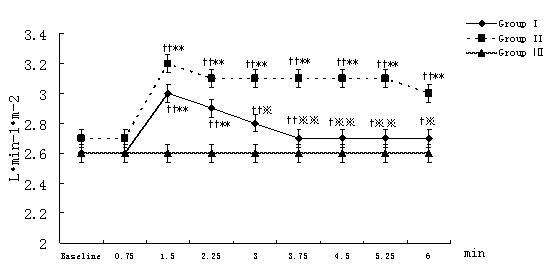
Figure 3. Changes in CI at different time points after the beginning of local infiltration
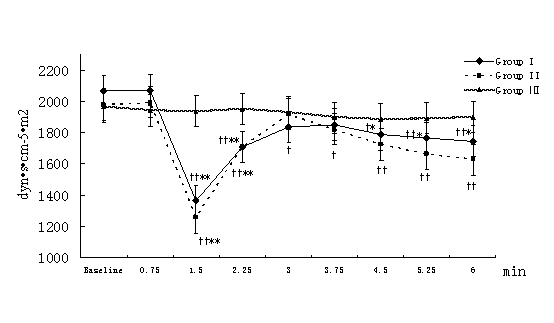
Figure 4. Changes in SVRI at different time points after the beginning of local infiltration
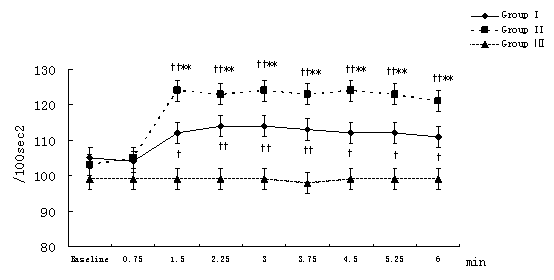
Figure 5. Changes in ACI at different time points after the beginning of local infiltration
Figure 1-5. Data are expressed as mean ± SD. The baseline hemodynamic values had no significant difference between Group I, Group II and Group Ⅲ. †P<0.05, ††P<0.01significant difference compared with baseline data within each group using repeated-measures ANOVA. ﹡P<0.05, ﹡﹡P<0.01 significant difference compared with data of group Ⅲ at the same time point in Group I or Group II using ANCOVA.. ※P<0.05, ※※P<0.01 significant difference compared with data of group II at the same time point in Group I using ANCOVA. MAP (mmHg) =mean arterial pressure; HR (bpm)=heart rate; CI (L·min-1·m-2)=cardiac index; SVRI (dyn·s·cm-5·m2) =systemic vascular resistance index; ACI(/100sec2) =acceleration index.
The average magnitude of hemodynamic changes compared with the baseline in group I and group II were as follows: MAP decrease>20%, SVRI decrease>35%, HR increase>15%, CI increase>15%, ACI increase>15%.(Figure 1-Figure 5)
Compared with group II at the same time point, group I had some significant differences: lower MAP (P<0.01 or P<0.05) at 2.25, 3, 3.75 minutes time point (In fact, compared with the baseline, MAP decreased in group I, while increased in group II) and lower CI (P<0.01 or P<0.05) at and from 3 minutes time point. (Figure 1)
DISCUSSION
Compression, electric coagulation, controlled hypotension, and local use of vasoconstrictors are commonly used to enhance hemostasis in FESS because of the rich vascularity of nose and the difficulty of bleeding control. Subcutaneous local infiltration of epinephrine is widely used to produce local vasoconstriction because of the direct effect on α-adrenergic receptors, thus will decrease surgical bleeding, lessen mucosal congestion and keep clear sight (1-5).
CI is determined by HR, preload, afterload, and contractility, while SVRI by afterload and ACI by contractility respectively (6-9). Significant hemodynamic changes happened after local infiltration in group I and group II. It was impossible to correlate these hemodynamic changes, including significant decrease in MAP and SVRI, and increase in HR, CI and ACI, with myocardial depression by general anesthetics, lidocaine and/or hypovolemia, because: 1) the volume of intravenous fluid infusion and the depth of general anesthesia before local infiltration remained almost constant in all the patients; 2) the local infiltration was applied 30 minutes or so after the induction and endotracheal intubations in all patients; 3) patients whose MAP fluctuation was above 10 mmHg in 3 minutes before local infiltration were excluded from our study; 4) increase in CI and ACI represented the enhanced myocardial function; 5) the last and most important reason, hemodynamic values in group Ⅲ had no significant changes at any time points in our study. In fact, many local anesthetics except cocaine have a biphasic effect on vascular smooth muscle; at low concentrations, these agents tend to cause vasoconstriction, whereas at clinically employed concentrations, they cause slight vasodilation without hypotension (4-5,10). So we concluded that hemodynamic changes were as a result of the absorption of epinephrine contained in lidocaine solution under general anesthesia.
The hemodynamic effects of epinephrine are dose-dependent and different dose epinephrine may activate different types of sympathetic receptors. A rate of 1 to 2µg/min, though rarely used, should predominantly activate β2-receptors with resulting vascular and bronchial smooth muscle relaxation. A rate of 2 to 10 µg/min should predominantly activate β1-receptors to increase heart rate, contractility, conduction and decrease the refractory period. Doses in excess of 10 µg/min cause marked α-stimulation with resultant generalized vasoconstriction (11).
Systemic absorption of epinephrine occurs when local infiltration is applied and systemic effects of epinephrine are variable in different patients and are related to its blood concentrations. Various studies have shown that the hemodynamic changes after local infiltration of epinephrine depend on physical status of the patient, epinephrine dose used, vascularity of the site of administration and its rate of absorption from the area infiltrated (4-5).
Nasal area has abundant vascularity and can absorb epinephrine quickly (12), so hemodynamic disturbances were observed from 1.5 minutes time point in group I and group II in our study. First, β2-receptor is most sensitive and β2-receptor-induced vasodilatation in muscle beds decreases system vascular resistance (SVR). That was why SVRI and MAP decreased at 1.5minutes time point. Few authors have reported anesthetics containing epinephrine-induced hypotension. For example, Murthy HS and colleagues (3) concluded that the combination of lidocaine and epinephrine might results in a biphasic hypotensive response around 2 minutes and 9–15minutes. Phillips S and colleagues (4) observed that arterial pressure decreased by more than 20% from preinfiltration values in 55% of patients who received local infiltration of epinephrine with lidocaine during neurosurgeries. Yang JJ and colleagues (5) concluded epinephrine contained in lidocaine could elicit a marked and temporary hypotension episode during FESS.
On the other hand, partial activation of β1-receptor elicited positive intropy, chronotropy and conduction in the heart, and caused increase in ACI and CI (which also associated with decrease of SVRI) at and from 1.5 minutes time point. Though CI increased, further reduction of blood flow to vital organs could be implied because of a distribution of blood to low resistance in the muscle. An increase in heart rate may be the baroreceptor reflex to a reduced blood pressure
Then MAP restored quickly from 2.25 minutes time point and reached the highest at 3minutes time point because of the utmost effect of β1- receptor and gentle stimulation on α-receptor, which could illustrate successfully why SVRI and ACI reached the highest values at 3 minutes time point either. John G and colleagues (12) reported patients during FESS showed a marked rise in plasma epinephrine concentration within 4 minutes of commencing infiltration. Homma Y and colleagues (13) also found mean plasma epinephrine concentration reached a maximum 3 minutes after administration of epinephrine during dental treatment.
Additionally, lower MAP at 2.25, 3, 3.75 minutes time point and lower CI at and from 3 minutes time point were observed when group I compared with group II, which implied that the hemodynamic changes induced by the absorption of epinephrine are dose-dependent.
Plasma epinephrine concentrations through vascular uptaking are relatively modest and are substantially less than the concentrations seen in psychologic stress and surgical stimuli (11). TIVA is widely used to offer a perfect general anesthesia in FESS (14-16). TIVA with TCI in our study provided a defined and deep general anesthesia (17) that may be a prerequisite for the induced-hypotension (5), because: 1) a deep general anesthesia reduced the production of endogenous catecholamines thereafter the changes in epinephrine blood concentration relative to endogenous epinephrine should be low in this study; 2) a deep general anesthesia covered the sympathomimetic effects of the total catecholamines in blood circulation. Additionally, deep general anesthesia may also decrease the bleeding, make endoscopic surgery technically easier, and improve endoscopic visualization of the surgical field (14-16).
Automated noninvasive blood pressure monitoring still has few limitations in clinical application. For example, it spends at least 30-35 seconds to measure arterial pressure. This is the major reason why MAP decrease in this study is much less than our previous study (5) (Average MAP decrease more than 28%) and why the epinephrine-induced hypotension is often neglected in clinics. Furthermore arterial pressure often can’t be measured in one time when arterial pressure changes too much. In our study, four patients were excluded because their arterial pressure couldn’t be measured in one time and the continuous two measurements missed the scheduled time point.
When anesthetists or surgeons who don’t know the above rule detect hypotension after infiltration, they might make wrong judgment, then wrong treatment including infusion of ephedrine or phenylephrine intravenously which obviously leads to marked and unnecessary hypertension when the blood concentration of epinephrine achieves the utmost. The correct way maybe is careful observation without any treatment because it only lasts a very short time (5).
There are some limitations in our study. First, to testify our viewpoints and illustrate this phenomenon more clearly, the plasma concentrations of catecholamines, particularly epinephrine and noradrenalin, should be measured. Second, the time frame of data collection was six minutes during which though the biggest hemodynamic changes including the lowest and the highest hemodynamic values were observed, we haven’t followed all the hemodynamic changes for a long enough period until all parameters returned to the baseline values.
According to the results of our study, we conclude that local infiltration with low dose epinephrine causes marked hemodynamic changes including decrease in MAP and SVRI, and increase in HR, CI and ACI during FESS under general anesthesia.