J Pharm Pharmaceut Sci (www.cspscanada.org) 9(1):32-39, 2006
Permeability enhancing
effects of the alkylglycoside, octylglucoside, on insulin permeation across
epithelial membrane in vitro.
Phani Prasanth Tirumalasetty, John G. Eley
Corium International, Inc.,
Department of Pharmaceutical Sciences,
Received 19 September 2005, Revised 17 January 2006, Accepted 18 January 2006, Published 2 February 2006
Corresponding Author:
John G. Eley, Department of Pharmaceutical Sciences,
Abstract: PURPOSE: To evaluate the permeability enhancing effects of octylglucoside (OG) for molecules with poor absorption such as insulin by in vitro cell models. METHODS: Transepithelial electrical resistance (TEER) was monitored to ensure monolayer integrity. Permeability was ascertained using paracellular markers. Markers and insulin were dissolved in Hanks balanced salt solution and placed on the apical side of the cells in Transwell® plates and allowed to diffuse under sink conditions. RESULTS: The effect of OG on the permeability of molecules across both monolayers was concentration and time dependent. Enhanced transport of the three molecules was observed across both monolayers treated with OG as compared to untreated monolayers. The effects of OG were reversible at low concentrations but there was permanent damage to cells at higher concentrations. Absorption enhancement was greater across T-84 monolayers compared to Caco-2 monolayers. CONCLUSIONS: The results indicate OG has potential as a permeability enhancer for poorly absorbed drugs with no significant damage to monolayers at low concentrations. Immediate attenuation in TEER upon exposure to OG indicates that permeability enhancing effects were likely to be associated with modulation of tight junctions suggesting the involvement of paracellular transport.
INTRODUCTION
Latest developments in biotechnology have led to the synthesis of a diverse array of peptide drugs (1). A major hindrance to deliver therapeutically effective doses of these macromolecules is their poor absorption through biological membranes leaving the parenteral route as the only choice for delivery. Because of the potent pharmacological effects and expected low toxicity of such drugs research is being directed to develop non-invasive routes of delivery (2).
These include transdermal, nasal, pulmonary, buccal and ocular (3). These routes are attractive because of the circumvention of first-pass metabolism and consequent route via endothelial membranes allowing rapid exchange of fluids and dissolved substances (4). However, these routes have limitations such as enzymatic breakdown and epithelial barriers, in view of which, several approaches have been investigated to improve bioavailability. One such approach is coadministration of transport enhancers and protease inhibitors (5, 6).
Absorption enhancers such as bile salts, surfactants, and phospholipids have been employed to improve bioavailability (7, 8). Although this approach has shown improved bioavailability, their mucosal toxicity has been a drawback, highlighting a requirement for non toxic permeation enhancers (9, 10).
Surfactants form a major group of paracellular
permeability enhancers (PPEs) which enhance the permeation of molecules across biological
barriers. Alkylglycosides are a class of water-soluble nonionic surfactants
consisting of a series of compounds with alkyl chain lengths of between 5 and
13 carbons glycosidically linked to a mono- or disaccharide. Orally
administered alkylglycosides are rapidly hydrolyzed to sugars and fatty
alcohols. (11). Octylglucoside (OG), an alkylglycoside, containing the
monosaccharide maltose ring, glycosidically linked to an 8-carbon alkyl chain
has been successful in improving absorption of drugs (12). A significant increase in the rectal absorption
of carboxyfluorescein in the presence of alkylglycosides having carbon chain
length of 8-12 carbons was observed concluding OG improved rectal absorption
(13). It has been reported that among the alkylglycosides studied,
dodecylmaltoside and OG were effective in lowering the resistance of human
carcinoma cell lines by opening tight junctions and allowing efficient cell
recovery (14).
The aim of this investigation was to evaluate the absorption enhancing effects of OG on the permeability of insulin across human carcinoma monolayers. The toxicity of OG was assessed by measuring the reversibility of transepithelial electrical resistance (TEER). Paracellular permeation effects of OG were characterized by studying the permeation of lucifer yellow and 3[H]-mannitol used as paracellular transport markers.
MATERIALS
AND METHODS
Hank’s balanced salt solution (HBSS), Dulbecco’s Modified
Eagle Medium (
Cell maintenance
Caco-2 and T-84 cells were employed to evaluate the effects of OG. These cells originate from human adenocarcinoma and exhibit characteristics of intestinal epithelium such as microvilli, intercellular tight junctions, enzymes, nutrient and efflux transporters and are appropriate models for evaluating the permeability of drug molecules (15, 16, 17).
Cells were seeded
at 1x104 cells/ml in 75cm2 flasks
until confluent. Caco-2 and T-84 cells were maintained in DMEM/High glucose and
Measurement of transepithelial electrical resistance (TEER)
Resistance
(Ohms.cm2) across cell monolayers was measured using a voltohmmeter at
24 hours intervals until and after cells attained confluence. Instrumentation
consisted of an Evom Epithelial voltohmmeter and an Endohm tissue resistance
measurement chamber (World Precision Instruments,
Preparation of solutions
Compounds were dissolved in HBSS at pH 7.4 except insulin, which was dissolved in 0.1N HCL (pH 2.3) then made up to the required quantity with HBSS to maintain physiological conditions (pH 7.4) during transport studies.
Transport studies
Transport studies were conducted using TranswellÒ plates at room temperature from the apical to basolateral side. 3[H]-mannitol, lucifer yellow and insulin were placed on the apical side at the following concentrations 0.1mCi/ml, 0.025%w/v and 0.067% w/v respectively with and without OG at concentrations of 0.2% - 0.5% w/v in HBSS and HBSS (1.5 ml) was placed in the receiver compartment. Inserts from TranswellsÒ were moved to wells containing fresh HBSS at predetermined intervals up to 6hr to maintain sink conditions. Samples collected from basolateral compartments were analyzed for Lucifer yellow and insulin by reversed phase high-pressure liquid chromatography (RP-HPLC). 3[H]-mannitol was determined by a scintillation counting using a Beckman LS 5000 TD and a LS 5000 TA series liquid scintillation system (Fullerton, CA).
HPLC Analysis
The HPLC system (Varian, CA) consisted of a Prostar 210/215 solvent delivery module connected to Prostar 320 UV/VIS detector and Prostar 400 Autosampler.
Chromatographic conditions for Lucifer Yellow:
A C-18 column (150 x 4.6 mm) with 5mm particles of 100 Å pore size (Microsorb, Varian, CA) was used in conjunction with a mixture of water/methanol (70:30) with paired ion chromatography (PIC) ‘A’ reagent as mobile phase. At room temperature, flow rate 1ml/min and at lmax 280 nm LY was eluted after 6 minutes.
Chromatographic conditions for Insulin:
A C-8 column (150x4.6mm) with 5mm particles of 100 Å pore size (
Scintillation
counting:
Concentration of 3[H]-mannitol transported to the basolateral side was detected by mixing 500ml aliquot of sample with 2 ml of Scintiverseâ and counted for 2 minutes.
Recovery studies
TranswellsÒ plates containing cells treated with OG were rinsed twice with HBSS. Both apical and basolateral chambers were replenished with fresh media and replaced in the incubator. TEER values were recorded every hour for 12 hours from the point of incubation to observe increase in TEER indicating cell recovery.
Data Analysis
TEER was measured prior to each experiment to ensure the confluency of the monolayers and also during transport studies to observe the effect of transport enhancer at each sample point. The apparent permeability coefficient (Peff) was calculated using the following equation.
Peff = (dQ/dt)*(1/A)*(1/C0).
Where dQ/dt (cumulative amount transported, μg/sec) is the flux of the marker/drug compound across Caco-2 cell monolayers and is measured as the slope of the regression line describing the cumulative amount versus time. A (cm2) represents the diffusional area of the inserts, and Co (μg/mL) denotes the initial concentration of marker/drug compound in the donor compartment. (20, 21). All measurements and P(eff) were in triplicate (n=3) and expressed as mean (±) SD values.
RESULTS
AND Discussion
Effect of OG on TEER of Caco-2 and T-84 monolayers
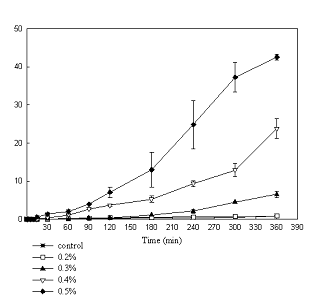
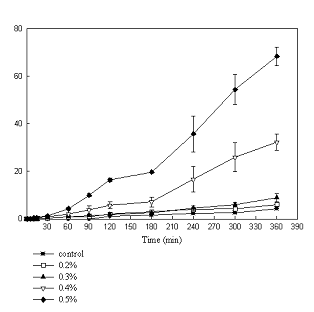
Previously thirteen alkylglycosides were evaluated for potential use as permeability enhancers using epithelial cells of which OG was found to be a promising candidate (14). In this present study, Caco-2 and T-84 monolayers were used to further evaluate the penetration enhancing effects of OG. To assess the effect of short term exposure both monolayers were treated with OG at 0.1% to 1% for 1 hour (data not shown). There was an immediate decrease in TEER compared to controls at these concentrations after 1 minute. At 0.1% after the initial fall, an increase in TEER was observed which could be due to low lipid to surfactant ratio resulting in surfactant binding to cell membrane causing stabilization (18). At higher concentrations, after an immediate fall, TEER continued to decrease over the exposure period. TEER was restored to its original value after removal of OG demonstrating a reversible effect when applied at 0.1% - 0.5% indicating optimum concentration range for this exposure time. The effect of OG also depends on time of exposure. The results using both monolayers exposed to OG for 6 hours are shown in figures 1 and 2. A concentration-dependent effect is evident on resistance across both monolayers showing an immediate fall followed by prolonged reduction.
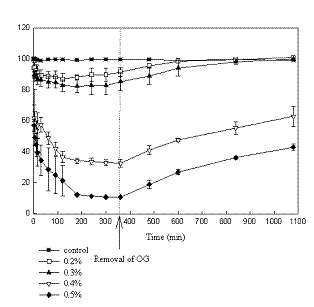
Figure 1. Effect of OG on TEER of Caco-2 monolayers for long exposure periods. TEER (%) vs. Time (mins)
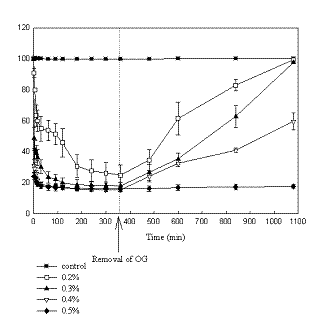
Figure 2.
Effect of OG on TEER of T-84
monolayers for long exposure periods. TEER (%) vs. Time (mins)
Both figures indicate that after a sharp fall, resistance continued to decrease over the six hour exposure period. The lower concentrations of 0.2% and 0.3% OG had less effect on Caco-2 cells than on T-84 cells. Higher concentrations exceeding 0.5% showed similar effects on both cells lines decreasing resistance by around eighty percent.
Figures 1 and 2 also illustrate the recovery of cells after removal of OG after 6 hours. Recovery is observed only when cells are incubated in culture medium containing serum indicating requirement of proteins for their repair and taking place over a period of 12 hours. Full recovery occurred in cells exposed to 0.2% and 0.3% OG and only partial or negative recovery for cells treated with higher concentrations. Lack of recovery indicates possible solubilization of tight junction associated proteins (TJAP’s) or ATP depletion which alters the phosphorylation state of TJAP’s thereby resulting in the inactivation of kinases (19). This was ascertained by microscopic studies revealing intercellular gaps between the cells and also detachment of cells from the monolayers exposed to concentrations ≥ 0.5% w/v. OG could possibly be affecting signaling pathways responsible for changing phosphorylation states of TJAP’s or myosin light chain components. The effect of OG was greater in T-84 than Caco-2 cells. This could be attributed to the well-developed junctional network in T-84 monolayers compared to discontinuous and poorly developed tight junctions in Caco-2 monolayers. This is evident from 4-5 times greater TEER values across T-84 monolayers. The minimum effective concentration of OG required to attenuate TEER over a period of 6 hours allowing recovery was found to be 0.2 -0.3% w/v. Higher concentrations disallow recovery. It is apparent that the effectiveness of OG in reducing resistance immediately upon exposure and allowing complete recovery after removal suggests a possible mode of action by the modulation of tight junctions associated proteins.
Effect of OG on the paracellular permeability
To determine if OG induced attenuation in TEER is accompanied by an increase in paracellular permeability, transport experiments were conducted using paracellular markers.
Effect of OG on permeability of paracellular markers
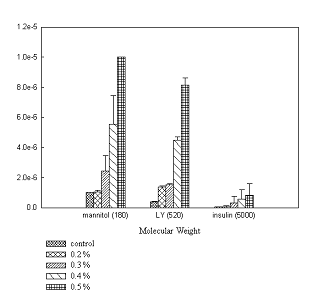
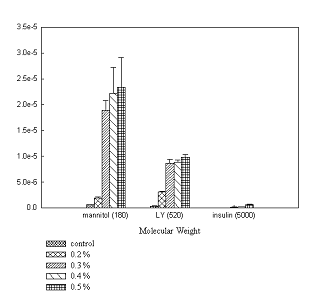
OG was applied in conjunction with paracellular markers to qualify its effect on paracellular permeability. In the absence of OG a low baseline permeability was observed with negligible amounts of both markers. The Peff values of markers across Caco-2 monolayers in controls was found to be in the same range as that observed by other investigators (20, 21). No significant difference in permeability of markers was observed at concentrations below 0.1% w/v compared to control. At concentrations ≥ 0.1%w/v, a noticeable improvement in the permeability of the marker was observed.
Tables 1 and 2 illustrate the cumulative amounts and permeability coefficient (Peff) of 3[H]-mannitol and lucifer yellow transported at 0.2%, 0.3%, 0.4% and 0.5% w/v concentrations of OG across Caco-2 and T-84 monolayers. The enhancing effect of OG on the transport of markers across both the cell lines is concentration dependent. The permeability of 3[H]-mannitol was greater compared to lucifer yellow across both monolayers attributed to the different physico-chemical properties of the markers such as the lower molecular weight of 3[H]-mannitol. The permeability of both markers was greater across T-84 than Caco-2 monolayers due to the more organized junctional complex in T-84. It has been proposed that resistance across monolayers is proportional to the number of strands found within tight junctions. Since OG is thought to affect tight junction proteins and T-84 cells contain more protein strands than Caco-2 cells, this results in higher permeability (22). Permeability of these markers indicates that OG induced decrease in TEER is associated with increased permeability across both monolayers.