J Pharm Pharmaceut Sci (www.cspscanada.org) 7(4):7-16, 2004
Molecular zipper assays: a simple homosandwich with the sensitivity of PCR.
Sujatha Guttikonda, Welson Wang, Mavanur Suresh1
Faculty of Pharmacy and Pharmaceutical Sciences, University of Alberta, Edmonton, Alberta, CanadaReceived 15 September 2004, Revised 16 September 2004, Accepted 20 November 2004, Published 12 January 2005.
PDF Version
Abstract
PURPOSE: The purpose of this study is to develop a simple and inexpensive method for detection of viral load or antigens present in the body fluids as for diagnosis or monitoring of infectious diseases. For example, in case of viral infection, nucleic acid based quantitative PCR/ RTPCR are sensitive in measuring viral load to follow the course of therapy or infection. The key limitations of such assays include the need for sample extraction, susceptibility to inhibitors, and high cost. METHODS: A molecular zipper assay based on the simple homosandwich concept for repeated epitopes was developed where the analyte or virus is sandwiched between the same antibodies for detection. A comparative study of the lower limit of detection of M13 model virus was performed with various substrates. RESULT: Homosandwich molecular zipper assay captured the model virus with high avidity resisting multiple rounds of washing. Detection of the virus by enzyme labeled MAb in combination with chemiluminescent substrates provided practical assay sensitivities of 7-15 phages and a theoretical detection sensitivity of one virus particle. CONCLUSION: The significance of our results on the molecular zipper assay relates to the development of ultrasensitive pathogen assays at low cost. Such assays could be developed for pathogenic bacteria and viruses, especially HIV & HCV viruses, which are ravaging impoverished continents of Africa, Asia and Latin America.
Introduction
The four major classes of pathogen diagnostics are culture tests, nucleic acid tests, antibody assays and antigen detection. The classical culture methods are confirmatory tests, but require considerable time for the final results. Nucleic acid detection (RNA or DNA), based on either target amplification or signal amplification is a powerful method which can theoretically detect a single copy of the infections agent. The chip-based detection method, multiplex PCR and immuno-PCR are some of the recent relatively complex approaches in the detection of viral antigens (1-3). Methodologies for detection of anti-pathogen antibodies are simple and robust, but fail to identify infections in patients who have not yet seroconverted and patients who are immunocompromised. These tests are generally indicative of exposure to a pathogen and often cannot distinguish between active infection and those who have recovered from the disease due to the slow clearance of antibodies. Detection of specific antigens of pathogens is a direct indication of infective states often detected by immunofluorescence, immunohistochemistry, agglutination assays and ELISA.
In this paper we describe an ultrasensitive molecular zipper ELISA as a routine monitoring and screening tool for viral load and as an inexpensive alternative to PCR. The concept is demonstrated using phage M13 model virus, since it is relatively safe and easily available. M13 is a filamentous phage with a major coat protein and few minor coat proteins (4, 5). The major coat protein is present in about 2700 copies per phage particle and is the product of gene VIII. Exploiting this architecture of M13 we employed the homosandwich assay to develop an ultrasensitive molecular zipper immunoassay concept with sensitivity comparable to quantitative PCR. A MAb directed against the M13 phage protein core was developed by traditional hybridoma technology (Liu. F., MSc thesis, University of Alberta, 1998). A bsMAb was also developed using the quadroma approach (6) with one paratope specific for M13 coat protein and the second paratope against alkaline phosphatase using the double fluorescent FACS sorting technique (7). In this two-step forward sandwich assay method, the solid phase antibody and the detector antibody are the same, thus eliminating the need for the second antibody with a different paratope as in traditional heterosandwich assays (8).
Materials and Methods
ELISA Reagents
The 96 well polystyrene plates from Nunc, USA were used for the ELISA. Anti-M13 antibodies were purified from the cell free-culture supernatant of P93 hybridoma in in vitro culture flasks or bioreactors (I-MAbTM , DCL, Canada). Biotinylation of P93 antibody was done using the long chain-sulfosuccinimidyl NHS biotin (Sigma, USA). Horseradish peroxidase (HRPO) and alkaline phosphatase (AP) are the two enzymes of choice for our experiments and the substrates used were tetramethyl benzidine (TMB) and para-nitrophenyl phosphatase (pNPP) for conventional assays, Quanta BluTM fluorogenic substrate (Pierce Endogen Inc., USA) for fluorescence assay and PicoTM or FemtoTM substrates (Pierce Endogen Inc., USA) for chemiluminescent assays.
Virus Purification
E. coli JM 109 bacteria streaked in M9 plate was used in bulk production of the M13 phages. The E. coli JM 109 was infected with M13 phages (10 ml10 ml JM109, 10 8 /ml & 200 ml of M13 solution, 10 8 / ml) in a 250 ml LB media for 5h at 37°C with vigorous shaking. Solution was centrifuged at 15000g for 20 min to remove the bacterial debris. 7% Polyethylene glycol-6000 (PEG-6000) and 2.3% NaCl (final) was added to the centrifuged solution and stirred gently. The purification of the phages was done by centrifuging the phage solution at 15000g for 40 min (9, 10). The pellet was resuspended in phosphate buffered saline pH 7.2 (PBS) and stored at -80°C. Traditional plaque assay to estimate virus titers was carried out by infecting 100 ml of JM109 with serially diluted M13 stock. The phage plus E. coli solution along with 3 ml of top agar was spread uniformly on the LB agar plate. Plaques were counted after incubating the plates at 37°C for 4-18 h.
Correlation of Plaque Titers to Epifluorescence Counting
Counting by epifluorescence microscopy using oligreen ssDNA quantitation reagent (Molecular probes Inc., USA) provided an independent, fast, efficient and inexpensive alternative to the transmission electron microscope method and also resulted in more precise and accurate estimates of virus (11-13).
Virus containing samples at various dilutions were stained with Oligreen ssDNA quantitation reagent (2 mg/ml in tris EDTA [TE] buffer pH 7.5) for at least 20 min at room temperature in a foil wrapped container. A 10 ml aliquot of the suspension was spotted onto a clean glass slide and 22mm2 cover glass (Fischer 11/2 ) was placed on the top. The slides were viewed with Zeiss LSM 510 confocal microscope under excitation/Emission at 500/520nm by using a 63X objective. Viruses were found to adsorb approximately equally to both the cover glass and the slide, and the value obtained was multiplied by 2 to account for the surface not counted in any one field (13). Commercially available FluoSpheresR - carboxylate modified microspheres, 1.0 mm diameter blue fluorescent (Molecular Probes, USA) were used as an additional guide to calibrate our epifluorescence counting. An aliquot containing 100 microspheres diluted from 3.6 x 1010 microspheres/ml stock solution was spotted on the slide and counted by epifluorescence.
Anti-M13 MAb Affinity Studies to M13 Phage Antigen
M13 virus (100 ml, 108 phages/ml in PBS) was coated on to the 96-well polystyrene plates. ELISA plates were blocked and washed with 200 ml of 1% BSA in PBS pH 7.2 and PBS-0.1% Tween 20 pH 7.2. Direct ELISA was performed by adding the biotinylated anti-M13 MAb concentrations ranging from 3.3nM - 667nM to the wells for 1h and washed three times with 0.1% PBS-Tween 20. Streptavidin-HRPO (SIGMA 1:10,000 dilution) was the reporter enzyme with TMB as the substrate for the final end point readings. Each value was done in quadruplicate and the experiment was repeated four times. Correlation coefficient and standard deviation were calculated to find the reliability of the Kd or Ka value. A double reciprocal plot with the inverse OD at 650nm against inverse biotinylated anti-M13 MAb concentration was plotted to estimate the affinity of the antibody towards the virus.
Molecular Zipper ELISA Using Monospecific Antibody
The anti-M13 capture antibody diluted in PBS buffer at 2 mg/well was immobilized on solid phase polystyrene 96 well microtiter plates for 3 h or at 4°C overnight. The plates were blocked with 200 ml of 1% dialysed- BSA (D-BSA, to remove traces of biotin) solution in PBS pH 7.2 by incubating for 2 h at RT. Appropriate dilutions of M13 were Incubated for 3 h at RT. To demonstrate the "molecular zipper effect" with high avidity binding of the virus to the antibody coated solid phase, the plate in some experiments were washed up to 15 times to dislodge the bound virus. Subsequently, 100 ml of biotinylated anti-M13 MAb (14) as "signal" antibody in 1% D-BSA solution in PBS at 0.15 mg/well was added. The plates were incubated for 3 h at RT followed by three washes with 0.1% PBS-Tween 20. Streptavidin-HRPO conjugate or streptavidin-AP (100 μl, 1:1000 dilution of stock) in 1% D-BSA solution was added and incubated for 1 h at RT and washed three times. To detect signal, 100 μl of various substrates were added (TMB, Quanta BluTM , PicoTM & FemtoTM for HRPO) and measured within 5-15 min at their respective wavelengths using Molecular Devices Gemini Spectramax XS microplate reader.
Molecular Zipper ELISA using Bispecific Antibody
The anti-M13 capture antibody at a concentration of 2 mg/well was immobilized on a solid phase polystyrene 96 well microtiter plates for 3 h or at 4°C overnight. The plates were blocked by incubating with 200 μl of 1% D- BSA solution in PBS pH 7.2 for 2 h at RT. A 100 ml aliquot of appropriate dilutions of M13 were added and incubated for 3 h at RT. A 100 ml of the bispecific MAb plus AP (anti-M13 X anti-AP) (15) at a concentration of 2.5 mg/well was added and incubated for another 1 h at RT. Each step was followed by an extensive washing three times with PBS-Tween 20. Conventional ELISA was performed with pNPP as the AP substrate. In the cyclic amplification procedure, a 50 ml aliquot of 0.2 M NADP in 0.9 M diethanolamine, pH 9.5, containing 1 mM MgCl2 was added to the wells and incubated for 15 min at RT. Then the amplification cycles were started by adding 110 l of cycling solution containing 50 U/ml alcohol dehydrogenase, 2.5 U/ml diaphorase and 0.55 mM iodonitotetrazolium in 25 mM sodium phosphate buffer, pH 7.2 including 4% (v/v) ethanol (15). The enzyme reaction was stopped after 15-20-minutes by 50 l of 0.4 M HCl. The plates were measured at 492 nm OD using an ELISA plate reader.
Statistics
Statistical analyses were performed using Sigma 2000 and Minitab 13.30 statistical software. Values were expressed as mean of hextuplicates or octuplicates. The practical detection of the analyte for each of the detection methods was determined by the student t-test of significance and p value with 95% confidence interval. The theoretical limit of detection was calculated from the mean of 20 blanks + 2SD. In all calculations, P<0.05 indicated a significant result.
Results
Virus Purification and Quantification
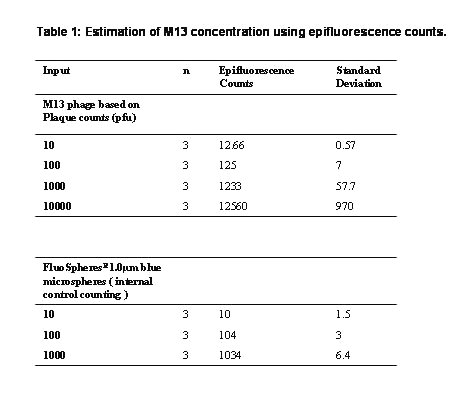
M13 virus provides a convenient, abundant and safe model pathogen for our ultrasensitive molecular zipper assay and is a filamentous bacteriophage which infects E. coli. We prepared a bulk culture of E. coli and M13 by established techniques (9) employing polyethylene glycol (PEG) precipitation and centrifugation to obtain a yield of ~ 1014 viruses/ml. Plaque assays on lawn of E. coli were initially used to determine the M13 titers. However it was necessary to quantify the phage stock by an independent method to investigate if the M13 purification procedures might have rendered some of the viruses non-infective by damage or loss of tail fibers. This is important because if the damage was significant, the plaque assays could artificially over estimate the real assay sensitivity at the ultrasensitive range of 1-100 virus particles if a substantial portion of the M13 is functionally defective in plaque formation, but yet could contribute to signal in the molecular zipper assay. Therefore, the plaque assay titre was correlated using the epifluorescence counting of M13 for correction of M13 titers. Oligreen ssDNA reagent (Molecular Probes, USA) stains only the single strand DNA (figure 1a) allowing the measurement of the fluorescent counts and provide a second method to estimate virus titers for correction of estimation obtained with plaque assay.
Figure 1: a Epifluorescence microscope study of M13 stained with Oligreen ssDNA quantitation reagent. Various dilutions of the virus stock were stained with Oligreen ssDNA quantitation reagent for 20 min and a 10 μl aliquot was spotted on the cover slip.
b Epifluorescence microscope results of E. coli JM109 stained with oligreen ssDNA quantitation reagent as a negative control.
In our results the discrepancy was ~17.6% (Table 1) with more viruses seen by epifluorescence at low levels of the analyte indicating some loss of infectivity by the purification procedure.
Table 1: Estimation of M13 concentration using epifluorescence counts.
We observed an error percent of 2.4 in our epifluorescent count estimation using FluoSpheresR - carboxylate modified 1.0 mm diameter blue fluorescent microspheres. E. Coli when used as a negative control is not stained by oligreen (figure 1b) but can be visualized with propidium iodide, acridine orange or gold nucleic acid stain that binds double stranded DNA (data omitted) . This result indicated that the Oligreen ssDNA reagent was specifically detecting M13 and not contaminating bacteria or their components.
The P93 anti-M13 antibody was purified by protein-G Sepharose chromatography of I-MAbTM bag based hybridoma bioreactor fluid. A typical harvest from a 4 week culture supernatant gave us ~ 10 mg of pure MAb. Affintiy study of the anti-M13 antibody to the solid phase M13 was done by an ELISA method (16). The affinity of the MAb to M13 was determined to be 4.7 X 10-9 M indicating that the anti-M13 MAb exhibits moderate apparent affinity.
The strength of binding is characterized by the affinity constant Ka or the dissociation constant Kd, which reflects the combination of the on-rate and off-rate of the antibody for M13 antigen. Since the IgG is bivalent and the M13 has numerous identical pVIII proteins, it is likely that many if not all the P93 may bind avidly with both paratopes. Hence our apparent affinity value could reflect a combination of both affinity and bivalent avidity.
Affinity of Anti-M13 Antibody to M13 Virus
Molecular Zipper Assay
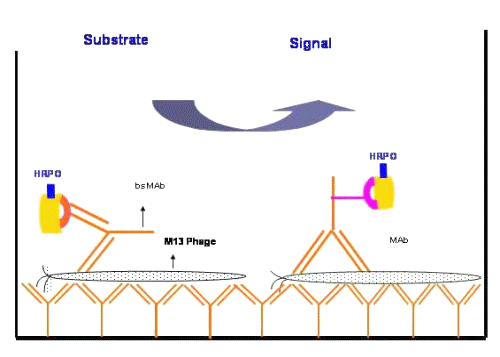
Diagrammatic representation of a "molecular zipper assay" is shown in figure 2.
Figure 2: Diagrammatic representation of the homosandwich format of molecular zipper ELISA assays. The solid phase is coated with bivalent monospecific MAbs capturing the M13 virus with multiple epitope-paratope interactions. On the distal side of the plate surface, monospecific or bispecific antibodies and various substrates are used in the various types of homosandwich assays.
Macro zippers are in everyday use and include the classical attachment of hairy seeds to the fibers of a cloth or the two ends of a zipper strap. The ability of the zippers to hold things strongly together stems from the numerous individual inter locking fibers as one unit, even though the strength of the individual interactions are weak. In a similar fashion we envisaged that an M13 virus particle with ~2700 identical repeat protein antigens could be captured by the numerous bivalent P93 MAbs coated on the microtitre plate. In this orientation, the filamentous M13 virus could be sequestered by a very high avidity interaction. One unexpected feature was observed during the first incubation of M13 with the solid phase coated with bivalent anti-M13 antibody illustrating the concept of our molecular zipper effect.
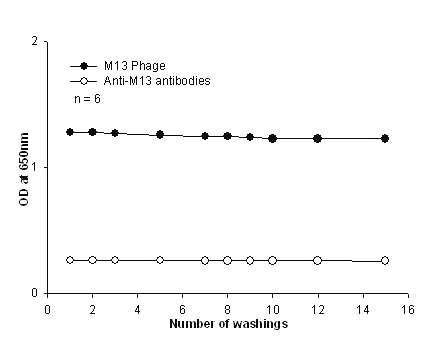
Multiple washings of the plate failed to dislodge the bound M13 virus from the antibody coated plate (figure 3).
Figure 3: Resistance to leaching of M13 phages and anti-M13 antibodies. The M13 was biotinylated and was added to the plate coated with anti-M13 antibody to observe virus leaching from the solid phase upon multiple washings. The firmly bound M13 was detected using streptavidin-HRPO. Similarly, biotinylated anti-M13 antibodies were added to the plate coated with 107 phages/well to observe the dissociation of the bivalent antibody. Washings were carried out up to 15 cycles and amount of leaching was measured by the difference in OD using TMB substrate. Each point was done in hextuplicates and plotted with error bars that are not visible since they are well below 5%.
After the 15th wash cycle, only 2-4% signal decrease was observed. In spite of the moderate affinity of the antibody, the antibody as a solid capture reagent was able to tightly bind the M13 virus and resist multiple rounds of washing. We believe this to be due to the "molecular zipper" effect. The virus has 2700 identical repeated coat protein epitopes resulting in the formation of multiple paratope-epitope interactions generating the high avidity entrapment of the virus even with the current random coating of the anti-M13 MAb on polystyrene plates. Thus the virus is held tightly to the solid phase by numerous individual moderate affinity interactions. Ordered covalent coating to orient the paratopes to from a 2-D array with even higher affinity antibody could likely generate a super molecular zipper for antigen capture at low concentrations of analyte to further increase assay sensitivity.
In addition, we also observed that the bivalent tracer antibody added to form the homosandwich also resisted multiple washings suggesting a potential involvement of both paratopes in binding to two adjacent pVIII protein epitopes (figure 3).
Detection Limits
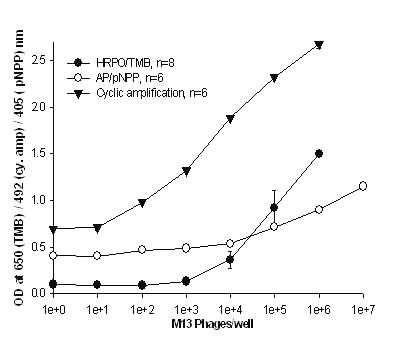
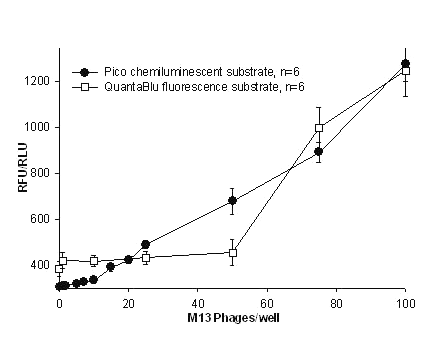
In our studies HRPO and AP were chosen as ELISA signal generating probes since they are the most common reporter enzymes in immunoassays. Practical detection limits by colorimetric methods using conventional TMB or pNPP for HRPO and AP respectively was in the range of 6,700 - 50,000 M13 particles (figure 4).
Figure 4: Detection of M13 using various substrates for detection sensitivity. The microtiter plate was coated with the P93 anti- M13 MAb. Following a wash various concentrations of M13 were added to the wells. The detecting antibody was biotinylated P93 MAb in cases where TMB and pNPP substrates were used. Anti-M13 X anti- AP, bsMAb was the detecting antibody for cyclic enzyme amplification method. This is the only assay in this study where the bsMAb has been employed. All the values are mean of hextuplicates except TMB which were octuplicates with their standard deviations.
The biotinylated MAb + streptavidin-AP in combination with pNPP gave the poorest sensitivity of 50,000 phage particles. The sensitivity of this assay was enhanced by using bsMAb and NADP based cyclic enzyme amplification (15, 17) and was observed to be 55 times better than colorimetric method with the detection limit at 125 phages/well (figure 4). This was the only instance that the bsMAb was used in this study. All other assays were using the monospecific MAbs.
Dramatic improvements in the sensitivity of M13 detection were observed with fluorogenic (QuantaBluTM, Pierce Endogen Inc) and chemiluminescent substrates (PicoTM & FemtoTM ) with detection limits of 75, 15 and 7 phages /well respectively (Table 2 & Figure 5 & 6).
Table 2: TMB and pNPP are conventional substrates, QuantaBluTM, PicoTM and FemtoTM are the fluorogenic and chemiluminescent substrates respectively from Pierce Endogen Inc. *Theoretical detection limits are extrapolated values from mean + 2 SD of 20 blanks.
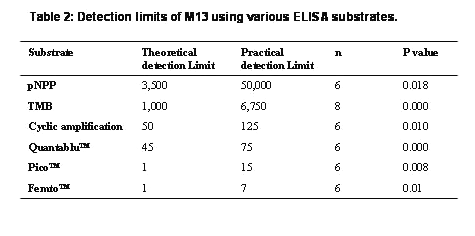
Figure 5: Detection of M13 using PicoTM chemiluminescent and QuantaBluTM fluorescent substrates for HRPO enzyme. The microtiter plate was immobilized with the P93 anti- M13 MAb. Following a wash various concentrations of M13 were added to the wells. The detecting antibody was biotinylated monospecific P93 MAb in both the assays. Streptavidin-HRPO conjugate was added which bound to the biotinylated MAb. All the values were mean of hextuplicates with their standard deviations.
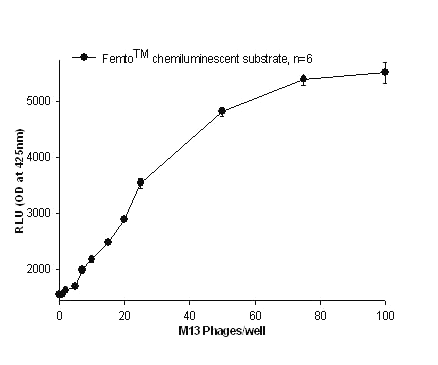
Figure 6: Detection of M13 using Femto TM chemiluminescent substrate for HRPO. P93 anti- M13 MAb immobilized microtiter plate was incubated with various concentrations of M13. The detecting antibody was again a biotinylated monospecific P93 MAb, to which streptavidin-HRPO conjugate was attached. All the values were mean of hextuplicates with their standard deviations.
A comparison of the two chemiluminescent substrates evaluated in this molecular zipper homosandwich M13 assay showed that FemtoTM chemiluminescent substrate was the most sensitive among all the substrates used with the practical detection limit at 7 phages/well (figure 6). The theoretical lower limit of detection based on the mean of twenty blank samples + 2 S.D gave a value of 1 virus particle for both Pico and Femto substrates (Table 2).
Overall the chemiluminescent ELISA assays were 1000 times more sensitive when compared to the conventional colorimetric assays. All the above homosandwich assays were performed using the monospecific antibody with the exception of the assay using cyclic amplification for signal generation.
Discussion
The strategy of virus detection using the forward two-site homosandwich molecular zipper immunoassay with the same monoclonal antibody has the advantage of ease of signal amplification by decorating the surface of the solid phase bound virus with multiple tracer antibody molecules. This is further amplified by the enzymatic reactions employing superior signal generating substrates.
Additional advantages of homosandwich assays include speed, specificity, economical viability, a lower susceptibility to several other influences seen in nucleic acid based PCR assays (18-21). An additional feature is that a larger measuring range could be obtained by appropriate choice of substrates in the same assay. By washing the plate and adding an alternative high or low sensitivity substrate if the primary ELISA is out of range, a wider range of analyte detection could be accomplished. The monoclonal antibodies labeled with biotin (21) are most versatile for immunoassays because they take advantage of the high affinity of biotin for the multivalent binding sites of streptavidin-labeled HRPO/AP. They also combine the amplification effect of biotin streptavidin interaction with high sensitivity of enzyme amplification employing fluorogenic or chemiluminescent detection methods (22). Bispecific antibodies with two non-identical paratopes ensure that every antibody molecule is uniformly bound to the tracer enzyme eliminating chemical manipulations generating the theoretical highest specific activity immunoprobe (23). However, since in this construct the paratopes are functionally univalent, avidity effects may not be as effective as bivalent antibodies.
This study emphasizes a comparison between the various new ultrasensitive substrates in the detection sensitivities of a virus. However, a detailed comparison of the relative merits of the monospecific and bispecific tracer has not been done since we are in the process of developing the anti-M13 x anti-HRPO bsMAb. Our results suggest a unique flexibility in estimation of the pathogens. This is accomplished by the judicious use of TMB or Femto TM substrate in the same microplate assay format. This approach could cover 6-8 log titers of viral load depending on a putative clinical sample tested. For example, a pathogenic virus (such as HIV or HCV) at an early stage of infection could have a lower load that could exponentially increase during late stages of the disease. An assay with a wide range would be useful in this situation. The theoretical limit of detection was estimated using 20 blanks +2 standard deviations and our results with the model virus show this theoretical sensitivity or LLD (lower limit of detection) approaches unity employing both chemiluminescent substrates (Table 2) even using monospecific andibodies. This corresponds to ~ 4300 yoctomoles of the viral antigen. Since each M13 has ~2,700 copies of the coat protein, the forward sandwich molecular zipper assay design allows the efficient capture and detection at ultra low levels with only one type of MAb.
Since the sensitivity of the assay is related to the affinity and specificity of the antibody, false negative or false positive determinations in a multiplexed molecular interaction based molecular zipper ELISA are less likely. Optimization of incubation time (24), signal antibody concentration, enzyme concentration, and plate pattern (25) was investigated by incubating the M13 antigens for 3, 5 and 18 hours with various concentrations of signal antibody and reporter enzymes. Increase in labeled MAb concentration or incubation time did not significantly increase the detection limits and only resulted in increased backgrounds (data not shown).
Our hypothesis is that the simple molecular zipper assay design can be extended to most viral and bacterial pathogens. It is based on the known fact that the architecture of most pathogens manifests the expression of multiple copies of identical surface epitopes (proteins, glycolipids, LPS etc.). This could be exploited to generate a high avidity entrapment on a solid phase for subsequent detection. Homosandwich assays for soluble antigens have been described (8) and this new adaptation has resulted in an unexpected avidity on the solid phase that could be used for diagnostics as well as pathogen isolation or clearance. The present assay format can potentially be applied to the detection of various medically important filamentous viruses as well as spherical viruses.
The molecular zipper effect is expected to be lower in the latter due to its geometry although more tracer MAb could bind instead to maintain sensitivity. The same strategy of homosandwich molecular zipper assays for repeating LPS epitopes of bacteria could also provide simple inexpensive assays for microbes and their spores. It is estimated that each E .coli bacterium has ~2x106 LPS molecules. A properly chosen antibody with low kinetic off rates (for good sequestration of the cell) and to a less critical invariant structural epitope could be effective in measuring both wild type and drug resistant strains.
The strategy of ultrasensitive virus detection using the molecular zipper homosandwich immunoassay has the advantage of combining three levels of amplification namely, avidity based capture, multiple decoration of distal epitopes and the use of sensitive substrates for signal enhancement by enzyme coupled reactions. Following the capture of the virus by the molecular zipper effect, decorating the surface of the solid phase bound virus on the distal epitopes with multiple tracer antibody molecules allows the formation of the homosandwich. It appears that the bivalent antibody binds with a higher apparent affinity (4.7 X 10-9 M) than the Fab fragment (3.5 X 10-8 M). Since the major coat protein of M13 has only 50 amino acids and few exposed epitopes (4) we believe that the higher apparent affinity for the bivalent MAb perhaps is due to binding with both arms. This is possible with the closely packed 2700 copies of the protein as a tubular array.
Our model study with M13 provided the most ideal conditions to demonstrate the fundamental concepts of a molecular zipper assay. However, we expect that in a clinical sample several additional issues need to be considered. Shed or free core/envelop antigens derived from pathogens could be a potential interference in whole pathogen assays that was not an issue in our M13 model study. A high capacity solid phase could however mitigate or eliminate this potential problem since the shed antigens are less likely to be part of the molecular zipper effect. Secondly, host antibodies against the pathogens would be present either in complexed or uncomplexed form. Hence, a prior treatment to inactivate this interference may be needed. Lastly unlike the M13 virus, many pathogenic mammalian viruses are enveloped viruses derived from host cell membrane. Consequently they would have fewer (~ few hundreds vs. 2700 for M13) surface viral envelope proteins. This could reduce the sensitivity of detection. A strategy for detection of these types of viruses could include the detergent disruption of the envelope to expose the nucleic acid + core protein complex, simulating the M13 phage.
Development and refinement of such molecular zippers could provide low cost immunoassays instead of the current expensive and complex PCR assays for a variety of medically important pathogens. A common architecture of viruses, bacteria and spores is the repeating surface structural determinants from a few hundred proteins or glycoproteins as in viruses (HIV & HCV) to more than a million LPS molecules on bacterial particles. However our molecular zipper assay strategy is not applicable to non-repeat epitopes.
Recently we have extended this molecular zipper concept to develop ultrasensitive detection of one to a few particles of E. coli 0157 (26), an enteropathogenic bacterium and Bordetella pertussis (27), one of the causative agents for whooping cough. While approaching the theoretical limit of immunodetection is a fundamental objective of our study, the strategy employed could provide the basis for developing cost effective assays for AIDS, hepatitis, and management of other diseases in underdeveloped countries and alleviating the strain on the health care budgets of developing countries.
Acknowledgements
MRS would like to thank the CBDN-NCE for operating grant, CHIR Industry award for salary support and Dr. David Bundle's lab for generous access to their Molecular Devices Gemini Spectramax XS microplate reader.
The authors would also like to thank Dr. D. Brocks for reviewing and suggesting changes to the manuscript.
References
Wang, Y. F., Shen, J. T. and Liu, H. H. Analytical performance of and real sample analysis with an HBV gene visual detection chip. Journal of Virological Methods, 121(1): 79-84, 2004.
Wang, N., Gao, X. Q. and Han, J. X. Simultaneous detection of HBV and HCV by multiplex PCR normalization. World Journal of Gastroenterology, 10(16): 2439-43, 2004.
Jane, K., Wright, N. and Morrow, R. Evaluation of quantitative and type-specific real time RT-PCR assays for detection of respiratory syncytial virus in respiratory specimens from children. Journal of Clinical Virology, 31: 123-129, 2004.
Henry, T.J. and Pratt, D. The proteins of Bacteriophage M13. Proceedings of National Academy of Sciences, 62: 800-807, 1969.
Bhardwaj, D., Singh, S. S., Abrol .S. and Chaudhary, V. K. Monoclonal antibodies against a minor and the major coat proteins of filamentous phage M13: their application in phage display. Journal of Immunological Methods, 179: 165-175, 1995.
Suresh, M. R., Cuello, A. C. and Milstein, C. Bispecific monoclonal antibodies from hybrid hybridomas. Methods in.Enzymology,121: 210-128, 1986.
Kreutz, F.T., Xu, D. and Suresh, M. R. A new method to generate quadromas by electrofusion and FACS sorting. Hybridoma, 17: 267-273, 1998.
Suresh, M. R. Immunoassays for cancer-associated carbohydrate antigens. In Longenecker BM eds. seminars in cancer biology- glycosylation changes associated with malignant changes Vol 2 W.D Saunders press p367-377, 1991.
Killington, R.A., Stokes, A. and Hierholzer, J.C. Virus purification. In Mahy. B.W.J and Kangro. H.O eds Virology Methods Manual. Academic press, London, p71-90, 1996.
Hermann, F. and Karlheinz, M. An electron microscopic study of bacteriophages from marine waters. Helgolander Meeresuntersuchungen, 41:385-414, 1987.
Weinbaurer, M. G.and Suttle, C. A. Comparison of epifluorescence and transmission electron microscopy for counting viruses in natural marine waters. Aquatic Microbial Ecology, 13: 225-232, 1997.
Hennes, K. P. and Suttle, A. Direct counts of viruses in natural waters and laboratory cultures by epifluorescence microscopy. Limnology and Oceanography, 40: 1050-1055, 1995.
Paul, H. J., Jiang, S.C. and Rose, J. B. Concentration of viruses and dissolved DNA from aquatic environments by vortex flow filtration. Applied and Environmental Microbiology, 57(8): 2197-2204, 1991.
Hermanson, G. T. Bioconjugate techniques Academic press, USA 1996.
Liu, F., Guttikonda, S. and Suresh, M. R. Bispecific monoclonal antibodies against a viral and an enzyme: utilities in ultrasensitive virus ELISA and phage display technology. Journal of Immunological Methods, 274: 115– 127, 2003.
Beatty, J. D., Beatty, B. G. and Vlahos, W. G. Measurement of monoclonal antibody affinity by non-competitive enzyme immunoassay. Journal of Immunological Methods, 100: 173-179, 1987.
Cook, D.B. and Self, C. H. Determination of one thousandth of an attamole (1 zeptomole) of alkaline phosphatase. Application in a immunoassay of proinsulin. Clnical Chemistry, 39: 965-971, 1993.
Martin, D., Arjona, A., Soto, I., Barquero, N., Viana, M. and Gomez-Lucia, E Comparative study of PCR as a direct assay and ELISA and AGID as indirect assays for the detection of bovine leukemia virus. Journal of Veterinary Medicine. Series B, 48: 97-106, 2001.
Greening, G. E., Woodfield, L. and Lewis, G. D. RT-PCR and Chemiluminescent ELISA for detection of enteroviruses. Journal of Virological Methods, 82: 157-166, 1999.
Starick, E., Ginter, A. and Coppe, P. ELISA and Direct immunofluorescent test to detect equine arteritis virus (EAV) using monoclonal antibody directed to the EAV-N protein. Journal of Veterinary Medicine. Series B, 48: 1-9, 2001.
Malergue, F. and Chungue, E. Rapid and sensitive streptavidin-biotin amplified fluorogenic enzyme-linked immunosorbent-assay for direct detection and identification of dengue viral antigens in serum. Journal of Medical Virology, 47: 43-47, 1995.
Porstmann, T. and Kiessig, S. T. Enzyme immunoassays techniques. Journal of Immunological Methods, 150: 5-21, 1992.
Cao, Y. and Suresh, M. R. Bispecific antibodies as novel bioconjugates. Bioconjugate Chemistry, 9: 635-644, 1998.
Sheppard, C., Cohen, B., Andrews, N. and Surridge, H. Development and evaluation of an antibody capture ELISA for detection of IgG to Epstein-Barr virus in oral fluid samples. Journal of Virological Methods, 93: 157-166, 2000.
Suli, J., Benisek, Z., Svrcek, S. and Ondreijkova, A. Correction of measurement errors in ELISA test. Bratisl Lek Listy,101(10): 565-568, 2000.
Guttikonda, S., Tang, X. L., Leung, K., Bhatnagar, P. and Suresh, M. R. E. coli O157 ultrasensitive detection. Immunology 2004. Medimond press Italy (in press) 2004.
Tang, X. L., Peppler, M. S., Irvin, R. T. and Suresh, M. R. Molecular Velcro Assays: Towards the theoretical limit of immunodetection of Bordetella pertussis employing bispecific antibodies. Clinical and Diagnostic laboratory Immunology, 11(4): 752-757, 2004.
Corresponding Author: Mavanur R. Suresh, 2118 Dentistry/Pharmacy Centre, University of Alberta, Edmonton, Alberta, Canada. msuresh@pharmacy.ualberta.ca.
Published by the Canadian Society for Pharmaceutical Sciences.
Copyright © 1998 by the Canadian Society for Pharmaceutical Sciences.
http://www.ualberta.ca/~csps