J Pharm Pharmaceut Sci (www.ualberta.ca/~csps) 7(3):303-309, 2004
High-performance liquid chromatographic determination of selected flavonols in Ginkgo biloba solid oral dosage forms.
Mary-Jean Dubber, Isadore Kanfer1
Division of Pharmaceutics, Faculty of Pharmacy, Rhodes University, Grahamstown, South AfricaReceived 19 July 2004, Revised 1 September 2004, Accepted 2 September, 2004, Published 24 September 2004.
PDF Version
Abstract
Purpose: This paper describes a validated high-performance liquid chromatographic (HPLC) - photodiode array (PDA) detection method to quantitate five flavonol components as markers; rutin, quercitrin, quercetin, kaempferol and isorhamnetin for use in the quality control of Ginkgo biloba dosage forms. Methods: Separation was achieved using a minibore Phenomenex Luna 5mm C18 (2) column with dimensions 250 x 2.00mm at 45°C with a one step linear gradient using acetonitrile:formic acid (0.3%) at a flow rate of 0.4ml/min. Results: The limits of quantitation for the flavonols were 2.76, 0.77, 1.11, 1.55 and 1.03mg/ml for rutin, quercitrin, quercetin, kaempferol and isorhamnetin, respectively. This method is linear over concentration ranges of 3-26mg/ml for all flavonols. Recoveries for rutin, quercitrin and kaempferol were above 94% while quercetin and isorhamnetin had average recoveries of 83% and 76%, respectively. Intraday precision did not exceed 6% and with-in day precision was better than 12% for all compounds. Conclusion: A suitable method was developed to identify and quantitate five relevant flavonol marker compounds and was successfully used to assay some commercially available solid oral dosage forms of Ginkgo biloba .
Introduction
Annual sales of botanicals and herbal remedies have been escalating universally with revenues in the USA alone exceeding $4.2 billion (1). Ginkgo biloba, commonly marketed as leaf extracts in pharmaceutical dosage forms, is currently one of the top selling herbal preparations with estimated worldwide sales in 1998 reaching a staggering $1 billion (2).
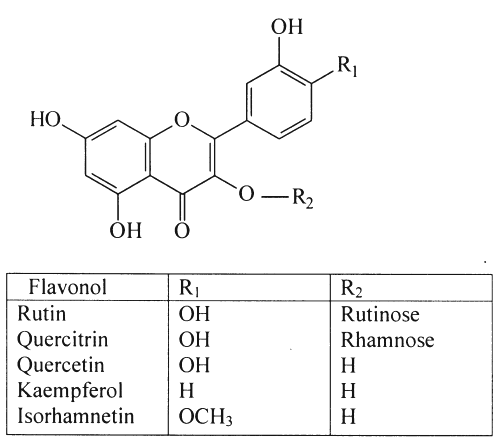
Ginkgo biloba's pharmacological activity has been linked to two major groups of compounds, the flavonoids and terpene trilactones (3-5). The flavonoids comprise of a large group of polyphenols that occur naturally in the plant kingdom (6,7) and include flavone and flavonol glycosides, acylated flavonol glycosides, biflavonoids, flavane-3-ols and proanthocyanidins (8). Of these, flavonol glycosides present in Ginkgo biloba leaves are more abundant than the other flavonoids. Numerous flavonol glycosides have been identified in Ginkgo biloba leaf extracts (9) as derivatives of the aglycones, quercetin, kaempferol and isorhamnetin that are present in the leaves in relatively small amounts (2). The flavonoids are collectively purported to be responsible for the free radical scavenging and antioxidant activity of Ginkgo biloba preparations (10-12). Recently, the flavonol glycosides have gained renewed interest and it has been suggested that absorption of flavonoids is not limited to the aglycone form (7,13). Structures of the aglycones, quercetin, kaempferol and isorhamnetin as well as 2 flavonol glycosides, rutin and quercitrin, are depicted in Figure 1.
Figure 1: Chemical structures of selected flavonols.
Another important group of Ginkgo components, the ginkgolides, are terpene trilactones, which are 20-carbon diterpene compounds with cage-like structures, only found in Ginkgo biloba . Five ginkgolides have been found: A, B, C, J and M as well as bilobalide, a 15-carbon sesquiterpene with a similar structure and is commonly grouped together with the terpenes. The ginkgolides have been shown to inhibit platelet-activating factor (PAF), an inflammatory autocoid (14).
Traditionally, the Chinese used brewed leaf extracts of Ginkgo biloba for the treatment of respiratory ailments such as asthma and bronchitis as well as for cardiovascular diseases (15). Additionally, extracts have been purported to be clinically useful for the treatment of various circulatory and demential disorders including Alzheimer's disease, depression, diabetic neuropathy, impotency, memory impairment, peripheral vascular disease, intermittent claudication, vertigo and tinnitus (16). However, the Cochrane Reviews (17, 18) indicate that such effectiveness is in doubt.
Currently, extracts are standardised to contain not less than 22.0% and not more than 27.0% flavonoids, calculated as flavonol glycosides and not less than 5.0% and not more than 7.0% terpene lactones (19).
In an effort to standardise Ginkgo preparations, various analytical techniques have been employed usually using various components as marker compounds. Flavonoid and terpene lactone content in Ginkgo provide useful criteria for product quality assessment (20).
Analysis of flavonoids with reversed phase HPLC was reviewed by Daigle and Conkerton as early as 1988. Ultraviolet (UV) detection was the most popular detection method due to the presence of strong flavonoid chromophores and availability of UV detection systems (21). Pietta and co-workers developed a binary HPLC gradient system applied to Ginkgo biloba for separation of naturally occurring flavonoids in 1991 (22) and Hasler et al. separated 33 flavonol glycosides found in Ginkgo leaves and extracts for fingerprinting (9) as well as quantification of aglycones after acid hydrolysis (23). In 1993, Sticher suggested using this approach to standardise phytomedicines containing flavonol components (24). Li and Fitzloff used HPLC with photodiode array (PDA) detection to compare flavonol aglycone content in various pharmaceutical products (3) and published a method to simultaneously determine terpene lactones and flavonol aglycones using HPLC with evaporative light scattering detection (ELSD) (25). Kressmann et al . calculated total flavonol glycoside content from hydrolysed flavonol aglycones to determine the pharmaceutical quality of several brands of Ginkgo biloba products available on the US market (26).
This paper describes a simple, precise, rapid and reproducible method to quantitate five relevant flavonol marker compounds in Ginkgo biloba dosage forms using HPLC. In addition to the usual markers, quercetin, kaempferol and isorhamnetin, separation and quantitation of the flavonol glycosides rutin and quercitrin have also been included. A simple and rapid extraction method using methanol deemed tedious sample clean-up unnecessary.
Methods
Chemicals and materials
Formic acid (analytical grade) was purchased from Merck (Darmstadt, Germany) and acetonitrile (HPLC grade) was obtained from BDH chemicals (Poole, UK). Rutin (95%) and quercetin (95%) were purchased from Sigma (Missouri, USA) and quercitrin (85%), kaempferol (90%) and isorhamnetin (99%) from Indofine Chemical Company (New Jersey, USA). The purity of these flavonols was assumed as provided by the suppliers and no adjustments were made in the quantitative analysis of the commercially available products. Water was purified in a Milli-Q system (Millipore, Bedford, MA, USA) and low protein binding Durapore (PVDF) filters were purchased from the same source.
Five Ginkgo biloba products (A, B, C, D and E) were purchased from a local pharmacy in Grahamstown, South Africa. Four of the products were solid oral dosage forms and one preparation contained herbal extract in a gelatin capsule.
Instrumentation
A Waters 2690 Separations Module and 2996 Waters Photodiode Array (Milford, MA, USA) equipped with an auto sampler, online degasser and column heater was used for HPLC analysis. Data were analysed and processed using the installed Empower Software. The flavonols were separated at 45°C on a minibore Phenomenex Luna 5mm C18 (2) column with dimensions 250 x 2.00mm using a one step linear gradient. Mobile phase A (acetonitrile) and B (0.3% formic acid) ratios where changed after 15 minutes from 15:85 to 25:75 and total run time was 33 minutes at a flow rate of 400ml/min.
Preparation of standard solutions
Separate stock solutions of the reference compounds were made by weighing the appropriate amounts of reference standards into volumetric flasks and filling them to volume using 50:50 methanol:water. Specific volumes of each solution relating to the required concentrations were pipetted into a 10ml volumetric flask and filled to volume with 50:50 methanol:water. This solution was used as the highest concentration of each component and serial dilution followed using 50:50 methanol:water for seven additional calibrators.
Sample Preparation and Extraction
Twenty five tablets of each solid oral dosage form were weighed and powdered in a mortar and pestle. A mass of powder equivalent to one tablet was weighed and 25 ml of methanol was added. The mixture was sonicated for a period of 1 hour, agitating the mixture manually after 30 minutes. The mixture was then centrifuged at 350 x g for 15 minutes and diluted with appropriate amounts of methanol and water in order to maintain a 50:50 methanol:water solvent ratio in all samples. The samples were then filtered through disposable 0.45mm PVDF filters before injection (5 ml).
Twenty five capsules (product C) were emptied and weighed and a mass of powder equivalent to the contents of a single capsule was extracted as previously described.
Method Validation
Calibration curves
Linear calibration curves were constructed on three consecutive days by analysis of a mixture containing each of the flavonols at four concentration levels and plotting peak area against the concentration of each reference standard. Calibration curves were constructed at the relevant wavelength of maximum absorption of each reference compound. Specificity was determined by calculation of peak purity facilitated by the photodiode array detector (PDA).
Precision and accuracy
The reproducibility of the assay was assessed by spiking tablet extracts (product A) with high, medium and low concentrations of each reference standard. Each concentration was prepared in triplicate over 3 days to determine both intra-assay and inter-assay precision.
Product A was chosen to perform recovery studies in triplicate and three blank extracts were prepared to determine the original concentration of chosen marker compounds.
A mass of powder equivalent to the mass of one tablet was spiked with low, medium and high concentrations based on the suitable range for each reference standard except for the low spike for rutin, since a significant amount of rutin was found to be present in this product.
Limits of detection (LOD) and quantitation (LOQ)
The limits of detection and quantitation were determined by means of serial dilution based on a signal to noise ratio of 3:1 for the limit of detection and 10:1 for the limit of quantitation.
Results and Discussion
Although various methods have been developed and published on the analysis of components in Ginkgo biloba dosage forms such as the flavonols and ginkgolides, those methods have either addressed the aglycone flavonols, quercetin, kaempferol (7) and isorhamnetin alone (2), ginkgolide components alone such as gingkolides A, B, C, J and bilobalide (15, 27,28) and more recently, simultaneous determination of some flavonol and ginkgolide components (25,29). Traditionally, the percentage of flavonol glycoside content was calculated from the aglycone content after acid hydrolysis. The total content of flavonol aglycones is then converted back to flavonol glycosides using molecular weight conversion factors. This approach has been widely accepted in the quality control of preparations containing flavonol components (2-4). Although this procedure is relatively simple and reference materials for the aglycones are available, true flavonol glycoside content is exaggerated. This is due to the intrinsic aglycones already present in Ginkgo biloba and calculations based on the average glycoside mass (2). In addition, glycosides are relatively stable compounds that hydrolyse only under rigorous extraction conditions and/or incorrect storage and are therefore excellent quality control indicators. An increase in the ratio of aglycones to glycosides in extracts signifies degradation (4). Although reference standards for all known flavonol glycosides are not available, relevant active markers amongst the flavonols could be chosen for analytical techniques to ensure comprehensive standardization.
Qualitative analysis of some flavonol glycosides in ginkgo leaf extracts has previously been investigated by both HPLC (2) and micellar electrokinetic capillary (MEKC) chromatography (30). Data from those studies were found to provide fingerprints to indicate adverse conditions of storage (2) since the presence/absence of rutin and other related glycosides such as quercitrin can serve as useful indicators of degradation (4).Quantitative analysis of flavonol glycosides which are relatively hydrophilic, have been largely overlooked since such compounds are usually poorly absorbed from the gastrointestinal tract following administration and thus not considered to contribute to clinical activity per se. Studies that are more recent have, however, indicated that flavonol glycosides are indeed more readily absorbed than their aglycone counterparts are and thus their presence/absence requires monitoring and assessment (31).
Linearity and range
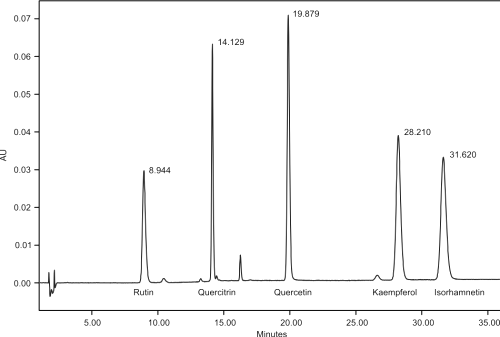
Figure 2 shows a typical HPLC-UV (350nm) chromatogram of five flavonols, rutin, quercitrin, quercetin, kaempferol and isorhamnetin with retention times 8.7, 14.1, 19.8, 28.1 and 31.5 minutes respectively.
The peaks were monitored in the UV range from 200-400nm. Quantitation was performed at 350nm for rutin, quercitrin and quercetin and 365nm for isorhamnetin and kaempferol, respectively. Assessment of peak purity showed peak homogeneity thereby excluding the possibility of the presence of interfering components and rendering the method specific. Linear ranges and correlation co-efficients are depicted in Table 1.
Figure 2: HPLC chromatogram of reference standards, rutin, quercitrin, quercetin, kaempferol and isorhamnetin with corresponding retention times at l = 350nm. Separation of the flavonols was achieved at 45°C on a minibore Phenomenex Luna 5mm C18 (2) column with dimensions 250 x 2.00mm using a one step linear gradient and flow rate of 400ml/min. Mobile phase A (acetonitrile) and B (0.3% formic acid) ratios where changed after 15 minutes from 15:85 to 25:75 and total run time was 33 minutes.
Table 1: Linear ranges and correlation coefficients of calibration curves (Day 1).
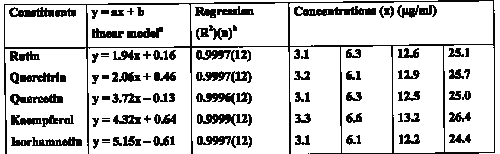
a y = peak area; x = concentration
b n = number of points in the calibration curve, each concentration injected 3 times, one at the start of the analysis, one in the middle and one injection after the tablet assay was completed.
Limits of detection (LOD) and limits of quantitation (LOQ)
Product E contained the highest quantities of marker compounds and was sequentially diluted to determine the LOD and LOQ in the tablet matrix. The limits of detection were 1.36, 0.34, 0.87, 0.90 and 0.75mg/ml for rutin, quercitrin, quercetin, kaempferol and isorhamnetin, respectively. The limits of quantitation were 2.76mg/ml for rutin, 0.77mg/ml for quercitrin, 1.11mg/ml for quercetin, 1.55mg/ml for kaempferol and 1.03mg/ml for isorhamnetin.
Precision and accuracy
The intra-assay precision represents data accumulated in triplicate over the first day of analysis. The relative standard deviations (% RSD) were less than 3% for all samples except for the low concentration of kaempferol (5.5%). The inter-day precision was calculated from nine determinations over 3 days for each concentration and was all below 8% except for isorhamnetin at both low and medium concentrations, which were 12.0% and 10.9%, respectively. Recovery data obtained from product A are depicted in Table 2.
Table 2: Recoveries of flavonols from Ginkgo biloba extract (product A).
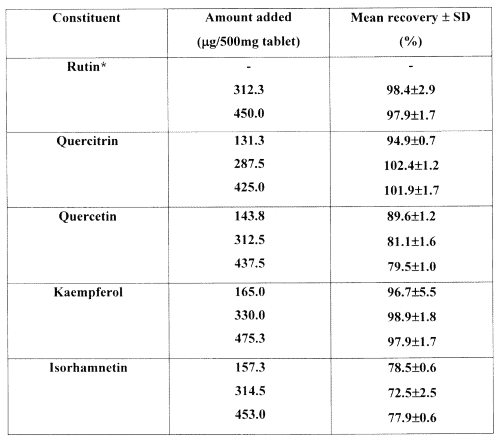
* Product A contained sufficient quantities of rutin to fall on the lower linear calibration range and therefore a low spike was not necessary.
Isorhamnetin showed consistent but somewhat lower recoveries at all three spiking levels compared to the other flavonols. Isorhamnetin has a methoxy functional group at the R 1 position (Figure 1) and exhibits lower polarity than the other marker compounds. A solvent for the sample solutions, chosen after an in-house investigation, proved that 50:50 methanol:water was best suited for use with the mobile phase. Low recoveries for isorhamnetin have been previously reported in both gas chromatographic-mass spectrometric (29) and HPLC analysis (32).
Tablet assay
Sample preparation and extraction of the solid oral dosage forms was simple and economical. Methanol was chosen as the extraction solvent due to its high polarity index, facilitating the extraction of both the polar glycosides (4) as well as the aglycone components, concurrently. Since the flavonol glycosides, rutin and quercitrin were also included in this analysis, sample hydrolysis was not required, conserving time and enabling rapid sample preparation.
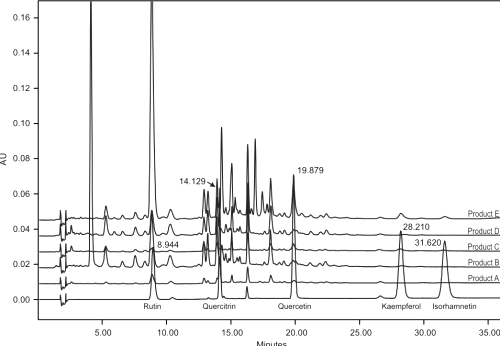
HPLC analysis of 5 Ginkgo biloba preparations gave remarkably similar fingerprint profiles (Figure 3) indicating higher amounts of rutin compared to the other flavonols.
Figure 3: Overlaid HPLC chromatograms of the reference standards including the profiles of the analysed Ginkgo biloba dosage forms (λ = 350nm).
This is in agreement with the literature where it was found that standardised leaf extracts contain only 0.39% (w/w) of kaempferol and 0.5% (w/w) quercetin [7]. Rutin as well as quercitrin and other related flavonol glycosides can serve as possible quality control indicators in Ginkgo products since changes in the ratio of rutin (and/or quercitrin etc.) to quercetin would indicate chemical change/degradation in the tablet matrix.
Quantitative analysis of five Ginkgo biloba dosage forms is shown in Table 3 over the 3 consecutive days.
Table 3: Flavonol marker content (per tablet) in 5 dosage forms purchased at a local pharmacy.
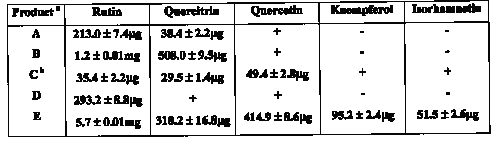
a n=9, analysis performed over 3 different days and all samples were prepared in triplicate
b content expressed per two tablets
(+) indicates that the marker could be identified but was below the LOQ
(-) indicates that the marker was not detected.
Average marker content in each of the brands varied markably. A 25-fold difference in the content of rutin was found between products A and E. Product E was the only product where all 5-marker compounds were found to be present in significantly higher amounts. Label claims gave no indication of individual marker content. Product B gave detailed information regarding amounts of total flavonol glycosides, ginkgolides and bilobalide, the remaining products simply specified the contents based on raw material incorporated into each tablet/capsule.
Conclusions
To date, quantification of flavonol glycosides, rutin and quercitrin in solid oral dosage forms have not been investigated. This paper includes the analysis of the flavonol glycosides, rutin and quercetrin as well as the aglycones, quercetin, kaempferol and isorhamnetin. It thus provides for the determination of rutin and quercitrin which can be useful indicators of stability and which is circumvented when samples are hydrolyzed to back calculate flavonol glycoside content. A simple, precise, rapid and reproducible RP-HPLC method was developed to quantitate these five flavonols as quality marker compounds with successful application to Ginkgo biloba dosage forms. Sample preparation was simple and no tedious cleanup was necessary. Product analysis showed similar qualitative profiles but remarkably different quantitative composition. These results indicate that suitable quality control measures need to be implemented to ensure the quality, safety and efficacy of commercially available Ginkgo biloba products.
Acknowledgements
Funding from the South African Medical Research Council is gratefully acknowledged.
References
Marcus, D.M. and Grollman, A.P., Drug therapy. N Engl J Med, 347:2073-2076, 2002.
Van Beek, T.A., Chemical analysis of Ginkgo biloba leaves and extracts. J Chromatogr A, 967:25-55, 2002.
Li, W., and. Fitzloff, J.F., HPLC determination of flavonoids and terpene lactones in commercial Ginkgo biloba products. J Liq Chrom Rel Technol, 25:2501-2514, 2002.
Sticher, O., Meier, B. and Hasler, A., The analysis of ginkgo flavonoids, in van Beek TA (ed), Ginkgo biloba. Harwood, Amsterdam, pp 179-202, 2000.
Nishida, S., and Satoh, H., Comparative vasodilating actions among terpenoids and flavonoids contained in Ginkgo biloba extract. Clin Chim Acta, 339:129-133, 2004.
Boue, S.M., Carter-Weintjies, C.H., Shih, B.Y. and Cleveland, T.E., Identification of flavone aglycones and glycosides in soybean pods by liquid chromatography-tandem mass spectrometry. J Chromatogr A, 991:61-68, 2003.
Watson, D.G. and Oliveira, E.J., Solid-phase extraction and gas chromatography-mass spectrometry determination of kaempferol and quercetin in human urine after consumption of Ginkgo biloba tablets. J Chromatogr B, 723:203-210, 1999.
Sticher, O., Ginkgo preparations: Biochemical, Pharmaceutical, and Medical Perspectives, in Kanba S: Richelson E (eds), Herbal Medicines for Neuropsychiatric Diseases. Seiwa Shoten Publishers, Tokyo, pp 83-114, 1999.
Hasler, A., Sticher, O. and Meier, B., Identification and determination of the flavonoids from Ginkgo biloba by high-performance liquid chromatography. J Chromatogr A, 605:41-48, 1992.
Stackman, R.W., Eckenstein, F., Frei, B., Kulhanek, D., Nowlin, J. and Quinn, J.F., Prevention of age-related spacial memory deficits in a transgenic mouse model of Alzheimer’s disease by chronic Ginkgo biloba treatment. Exp Neurol,184:510-520, 2003.
Ellnain-Wojtaszek, M., Kruczynski, Z. and Krasprzak, J., Investigation of the free radical scavenging activity of Ginkgo biloba leaves. Fitoterapia, 74:1-6, 2003.
Goh, L.M., Barlow, P.J. and Yong, C.S., Examination of antioxidant activity of Ginkgo biloba leaf infusions. Food Chem, 82:275-282, 2003.
Goh, L.M.L. and Barlow, P.J., Flavonoid recovery and stability from Ginkgo biloba subjected to a simulated digestion process. Food Chem, 74:195-202, 2004.
Chavez, M.L. and Chavez, P.I., Ginkgo (part 1): history, use, and pharmacologic properties. Hosp Pharm, 33:658-672, 1998.
Jensen, A.G., Ndjoko, K.,Wolfender, J-L., Hossettmann, K., Camponovo, F. and Soldati, F., Liquid chromatography-atmospheric pressure chemical ionisation/mass spectrometry: a rapid and selective method for the quantitative determination of ginkgolides and bilobalide in ginkgo leaf extracts and phytopharmaceuticals. Phytochem Anal, 13:31-39, 2002.
Chavez, M.L. and Chavez, P.I., Ginkgo (part 2) clinical efficacy, dosage, and adverse effects. Hosp Pharm, 33:1076-1095, 1998.
Evans, J.R., Ginkgo biloba for age-related macular degeneration. Cochrane Database Syst Rev, 2:CD001775, 2000.
Birks, J., Grimley, E.V. and van Dongen, M., Ginkgo biloba for cognitive impairment and dementia. Cochrane Database Syst Rev, 4:CD003120, 2002.
German Pharmacopoeia. Deutscher Apotheker Verlag, Stuttgart, 2000.
Busse, W.R., The role of markers for the quality determination of botanical raw materials and extracts. AAPS Dietary Supplements Forum, Washington DC, June 28-30, 2000.
Daigle D.J. and Conkerton, E.J., Analysis of flavonoids by HPLC: an update. J Liq Chromatogr, 11:309-325, 1988.
Pietta, P., Mauri, P. and Bruno, A. Identification of flavonoids from Ginkgo biloba, Anthemis nobilis and Equisetum arvense by high-performance liquid chromatography with diode-array UV detection. J Chromatogr A, 553:223-231, 1991.
Hasler, A., Sticher, O. and Meier, B.J., High-performance liquid chromatographic determination of five widespread flavonoid aglycones. J Chromatogr, 508:236-240, 1990.
Sticher, O., Quality of Ginkgo preparations. Planta Med, 59:2-11, 1993.
Li, W. and Fitzloff, J.F., Simultaneous determination of terpene lactones and flavonoid aglycones in Ginkgo biloba by high-performance liquid chromatography with evaporative light scattering detection. Pharm Biomed Anal, 30:67-75, 2002
Kressmann, S., Muller, W.E. and Blume, J., Pharmaceutical quality of different Ginkgo biloba brands. Pharm Pharmacol, 54:661-669, 2002.
Ganzera, M., Zhao, J. and Khan, I.A., Analysis of terpene lactones by high-performance liquid chromatography and evaporative light scattering detection. Chem Pharm Bull, 49:1170-1173, 2001.
Lang, Q., Kwang, H. and Wai, C.M., Selective dissolution and one step separation of terpene trilactones in ginkgo leaf extracts for GC-FID determination. Talan, 54:673-680, 2001.
Deng, F. and Zito, S.W., Development and validation of a gas chromatographic-mass spectrometric method for simultaneous identification and quantification of marker compounds including bilobalide, ginkgolides and flavonoids in Ginkgo biloba extract and pharmaceutical preparations. J Chromatogr A, 986:121-127, 2003.
Pietta, P.G. and Mauri, P.L., Application of micellar capillary chromatography to the determination of flavonoid drugs. J Chromatogr, 549:367-373, 1991.
Hollman, P.C.H. and Katan, M.B., Dietary flavonoids: intake, health effects and bioavailability. Food Chem Tox, 37:937-942, 1999.
Chin, L., Lin, Y.R., Huang, C.Y. and Wen, K.C., Evaluation of quantitative analysis of flavonoid aglycones in Ginkgo biloba extract and its products. J Food Drug Anal, 8:289-296, 2000.
Corresponding Author: I. Kanfer, Division of Pharmaceutics, Faculty of Pharmacy, Rhodes University, Grahamstown, 6140, South Africa. I.Kanfer@ru.ac.za
Published by the Canadian Society for Pharmaceutical Sciences.
Copyright © 1998 by the Canadian Society for Pharmaceutical Sciences.
http://www.ualberta.ca/~csps