J Pharm Pharmaceut Sci (www.ualberta.ca/~csps) 7(3):332-336, 2004
COX-2 selective inhibitors cardiac toxicity: getting to the heart of the matter.
Neal M. Davies and Fakhreddin Jamali1
College of Pharmacy, Washington State University, Pullman, Washington, USA and Faculty of Pharmacy & Pharmaceutical Sciences, University of Alberta, Edmonton, Alberta, CanadaReceived 11 October 2004, Revised 19 October 2004, Accepted 20 October 2004, Published 29 October 2004
PDF Version
Abstract
On September 30, 2004, Merck and Co. voluntary withdrew rofecoxib (Vioxx) from the market due to increased risk of cardiovascular events associated with the drug. This raised some issues. It is unclear whether the cardiac toxicities associated with rofecoxib isare due to its high COX-2 selectivity. Rofecoxib is the most specific COX-2 inhibitor among the first generation of the class, i.e., negligible COX-1 inhibitory effect. In addition to the gastrointestinal side effects, COX-1 inhibition is known to offer cardioprotection. This is one of the main present indications of aspirin-like drugs. It is plausible that the COX-2 inhibition is associated with altered homeostasis that is compensated with the cardioprotection effect of COX-1 inhibition that patients receive either through the less COX-2 selectivity of other NSAIDs or through co-administration of low dose aspirin. In addition, the cardiac toxicity of rofecoxib could be due to its unique chemical structure, its pharmacokinetics and tissue distribution, and/or the presence of toxic metabolites. Nevertheless, one cannot ignore the public need for NSAIDs with less gastrointestinal side effects than the traditional drugs. However, based on some available indirect evidence, and unless more clear-cut data become available, the use of highly COX-2 selective NSAIDs without the use of a suitable COX-1 inhibitor, (e.g., low dose aspirin) may be best avoided. This may be particularly relevant to the chronic use of these drugs.
On September 30, 2004 Merck and Co. instituted an immediate voluntary worldwide withdrawal of Vioxx (rofecoxib) a selective inhibitor of cyclooxygnease-2 (COX-2). Rofecoxib was approved by the United States Food and Drug Administration (FDA) in May 1999, accounted for $2.5 billion in worldwide sales in 2003. It was indicated for osteoarthritis, rheumatoid arthritis, severe menstrual cramps and, in higher dose-strengths, for short-term relief of acute pain. It was recently approved for use in children with juvenile rheumatoid arthritis. This is the first prescription drug since 2001 to be taken off the market for safety reasons. However, there is a long history of the removal of non-steroidal anti-inflammatory drugs (NSAIDs) from the market for a variety of reasons. They include benoxaprofen, pirprofen, carprofen, bromfenac and a controlled-release formulation of indomethacin.
The regulatory approval of Vioxx was based on a safety database of Phase III studies; which included approximately 5000 patients on rofecoxib. The data did not show an increased risk of heart attack or stroke. Subsequently a double blind randomized, stratified, parallel group prospective clinical trial, VIGOR (VIOXX GI Outcomes Research), was conducted, with 8076 patients to compare the occurrence of gastrointestinal toxicity of rofecoxib (50 mg daily) versus another NSAID, naproxen (1000 mg daily), during chronic treatment for patients with rheumatoid arthritis (1). This study was primarily designed to examine gastro-intestinal side effects of rofecoxib. Aspirin use was not permitted in the study. The VIGOR study demonstrated that patients taking rofecoxib had fewer stomach ulcers and bleeding than patients taking naproxen, however, the study also showed a greater number of heart attacks in patients taking rofecoxib (1).
The incidence of myocardial infarction was lower among patients in the naproxen group than among those in the rofecoxib group (0.1 percent vs. 0.4 percent). Consequently, new safety information from the VIGOR study was added to the labelling for Vioxx in April 2002 that contraindicated using rofecoxib in obvious cases of ischemic heart disease. Changes to the summary of product characteristics and patient information leaflets highlighted the need for caution in high-risk patients. Subsequently, additional published data from other randomized controlled trials, retrospective observational studies, and spontaneous reporting schemes provided a conflicting body of evidence on the cardiovascular risk with COX-2 inhibitors. Some of these reports showed no differences in adverse cardiac events between COX-2 inhibitors and non-selective NSAIDs (2-4).
In 2000, Merck and Co. began a long-term prospective, randomized, placebo-controlled, double blind, multi-center clinical trial to test rofecoxib in adenomatous polyposis prevention (APPROVe). This trial provided an even more comprehensive picture of the cardiovascular safety profile of rofecoxib. Although there were some cardiovascular events within the first 18 months of treatment, these data did not show any increased risk of confirmed cardiovascular events in patients receiving rofecoxib. The main impetus for the withdrawal of rofecoxib, however, is suggested to be the results and analysis of the first three-year data. . In 2600 patients, rofecoxib 25 mg was compared with placebo in the prevention of the recurrence of adenomatous polyps of the large bowel in patients with a history of colorectal adenomas. The study included patients aged 40-96; approximately 62% male. In total, 25 patients receiving placebo and 45 receiving rofecoxib demonstrated thromboembolic events. There were three absolute event rates per 400 patient years in the placebo group versus six events per 400 patient years in the rofecoxib treatment group. These increased risk of confirmed serious thromboembolic events including heart attack and stroke appeared statistically evident at 18 months of chronic dosing. Preliminary data released showed that five people died out of the 1,287 taking rofecoxib during the almost three-year study, one from a heart attack and three from sudden cardiac death (i.e. conduction problems with the heart). One death was unrelated to the study. Among the 1,299 participants given a placebo, there were also five deaths - three from heart attacks, one from sudden cardiac death and one from another cause. There were no stroke deaths, although patients on rofecoxib had double the number of strokes as compared with those in the non-drug group. Although the number of deaths was equal in both groups, those on placebo had only 25 cardiovascular problems, compared to the 45 among those administered rofecoxib once a day. These findings, observed just two weeks from the end of the trial, suggested that rofecoxib doubled the risk of heart attack or stroke.
In contrast with the VIGOR trail, aspirin use<325 mg per day was permitted during the APPROVe study. However, aspirin was not administered as part of the protocol, and salicylate or thromboxane concentrations were not monitored during the study. It has been suggested that the use of concomitant ASA did not have a significant difference in the cardiac toxicity profile of the APPROVe patients, however, the objective of the study did not include the systematic study of ASA's effects(5).
With the voluntary withdrawal from the market of rofecoxib, the biomedical community is left with several unanswered questions that need to be addressed:
Are the cardiac toxicities associated with rofecoxib due to COX-2 specificity?
There are several other COX-2 inhibitors similar to rofecoxib (Vioxx) on the market including: Celecoxib (Celebrex) approved in 1999, and a second generation of these agents with improved COX-2 selectivity have also been developed including valdecoxib (Bextra, approved in 2001), etoricoxib (Arcoxia), and lumiracoxib (Prexige), parecoxib (Dynastat), deracoxib (Deramaxx) (for veterinary use), tiracoxib and cimicoxib. Several other coxibs are currently undergoing clinical development. Meloxicam (Mobicox), an off-patent NSAID, has been demonstrated to be COX-2 selective.
Both the conventional and COX-2 selective NSAIDs elicit their beneficial anti-inflammatory effect through inhibition of the COX-2 pathway. Indeed the efficacy or potency of the anti-inflammatory effect of NSAIDs is independent of their degree of COX-2 selectivity. The conventional NSAIDs however, inhibit the COX-1 pathway as well. This pathway is responsible for both the main side effect of NSAIDs i.e. gastrointestinal damages, as well as their aspirin-like cardiovascular protection properties. Among the first generation COX-2 selective inhibitors (celecoxib and meloxicam) rofecoxib is the most selective one (Table 1).
Table 1: A selected group of non-steroidal anti-inflammatory drugs with various degree of selectivity in inhibiting the cyclooxygenase 2 pathway.
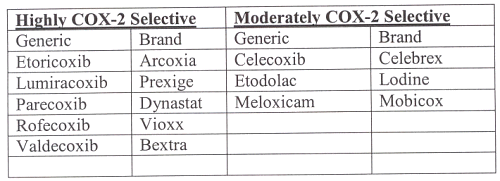
This was thought to be the main advantage of rofecoxib over other COX-2 selective NSAIDs. Indeed, all of the second-generation COX-2 selective inhibitors are designed to be even more selective than rofecoxib.
The rationale for developing highly selective COX-2 selective drugs is that, since the discovery of heterogeneity of COX enzymes, it has been believed that under healthy conditions, the COX-2 pathway is non-existent and it is only induced in response to an inflammatory assault. Conversely, COX-1 enzymes are constitutive and are required for the maintenance of the cell integrity (5). Hence, the goal of the molecular designers became to make NSAIDs with potent COX-2 but without COX-1 inhibitory effect. With this trend of thought two main assumptions were taken for granted: 1) The COX-2 pathway has no physiological role because it is non-existent under healthy conditions and 2) The lower the COX-1 activity, the safer the NSAID. Both these assumptions, however, are open to debate.
Firstly, more recent data indicate that COX-2 is present in healthy organs such as kidneys and brain (6) and appears to have physiological roles such as being involved in the electrolyte retention and hence may influence blood pressure (7). Secondly, COX-1 inhibition has cardiovascular protection properties; hence, highly COX-2 selective NSAIDs lack this important beneficial effect.
It is well acknowledged that patients with inflammatory conditions, more specifically those with rheumatoid arthritis, are more prone to cardiovascular complications than the general population (9). Most of these patients are usually under therapy with NSAIDs. Traditionally, they were treated with drugs with both COX-1 and COX-2 inhibitory activity. Furthermore, celecoxib and meloxicam also contain some appreciable COX-1 inhibitory activity (Table 1). Beneficial and adverse consequences of inhibition of each COX pathway in the absence of the other are not known. However, COX-2 inhibition appears to be not quite without physiological consequences at the cardiovascular level. This plausible effect, however, may be compensated by protective properties mediated by COX-1. The mixed effect is attained when either the NSAID possesses some COX-1 inhibitory activity or when aspirin (which irreversibly acetylates COX-1 and 2) is allowed in the regimen. The Celecoxib Long-Term Arthritis Safety Study (CLASS) was a double-blind, randomized controlled trial of 8059 patients, in which patients received celecoxib (400 mg twice per day), ibuprofen (800 mg three times per day), or diclofenac (75 mg twice per day) (10). The CLASS trial with celecoxib did not raise any alarm regarding significant differences in cardiovascular events as compared with other NSAIDs. However, Pfizer will begin a clinical trial of osteoarthritis patients with a history of heart disease to examine the efficacy of celecoxib against a placebo in terms of prevention and protection from heart attack and stroke (8).
Interestingly in the Vioxx trail (VIGOR) aspirin was not allowed but in the APPROVe trial <325 mg per day) was permitted although this was not systematically regulated or studied in detail.
Does pharmacokinetics and metabolism of rofecoxib play a role in this cardiac toxicity?
Although COX-2 inhibitors share similar mechanisms of action there are differences in their chemical structure and physicochemical properties. The pharmacokinetics of each COX-2 inhibitor is unique (11-12). Rofecoxib is metabolized primarily by cytosolic enzymes rather than cytochrome P450 to at least five metabolites. There is a paucity of information surrounding the activity or potential toxicity of these metabolites (12).
Our own unpublished data suggest that, at least in the rat, rofecoxib tends to be concentrated in the kidneys by many fold more than meloxicam. A positive correlation has also been observed between plasma concentration and sodium retention for rofecoxib but not for meloxicam. These animal data suggest important differences between two COX-2 selective NSAIDs in addition to their differences in their degree of COX-2 selectivity. Unfortunately, plasma concentration data were unavailable on the parent compound or metabolites in the patients in the APPROVe study.
In recent editorials on COX-2 inhibitors and cardiac toxicity the importance of pharmacokinetics in the action of COX-2 inhibitors has been inexplicably lacking. For instance, the disposition kinetics of rofecoxib and celecoxib are very different in every major pharmacokinetic parameter (11-12). Further studies aimed at optimizing the pharmacokinetic/pharmacodynamics of COX-2 inhibitors and the relevance of these pharmacokinetic parameters on the adverse effect profile are required.
Should the cardiovascular side effects of rofecoxib be extrapolated to all COX-2 selective NSAIDs?
In addition to being the most COX-2 selective first generation COX-2 inhibitor, rofecoxib has its own unique chemical structure. Hence, it is possible that these side effects are specific to this particular chemical structure. Unique side effects have previously been observed from one member of a drug class including NSAIDs. For example, benoxaprofen was withdrawn from the market due to its unique hepatic side effect.
Conclusions
The discovery of cyclooxgenase isoforms and subsequent research, which is in part supported by pharmaceutical companies, has lead to major scientific advances in the understanding of inflammation. The pathological and physiological roles of these COX isoforms still require further elucidation. Selective COX-2 inhibitors were developed due to a medical and societal need for safer anti-inflammatory drugs with respect to their gastrointestinal side effects. A careful prospective examination of rofecoxib, the most selective first generation COX-2 inhibitor, has revealed unexpected cardiovascular adverse effects. It is not known whether scrutinized regulated co-administration of low dose aspirin to all the patients in the APPROVe trial could have obviated the reported problems. Nevertheless, the lack of appreciable COX-1 inhibitory effect is a plausible explanation for the observed increased cardiovascular side effect associated with rofecoxib, although pharmacokinetic explanations cannot be discounted.
One cannot ignore the public need for NSAIDs with less gastrointestinal side effects than the traditional drugs. However, based on the rationale put forward, and unless more clear-cut data become available, the use of highly COX-2 selective NSAIDs (Table 1) without the use of a suitable COX-1 inhibitor, (e.g., low dose aspirin) may be best avoided. This may be particularly relevant to the chronic use of NSAIDs.
It should be borne in mind that many of the non-selective NSAIDs on the market have not been subjected to a long-term clinical trial such as APPROVe; hence their cardiovascular safety is not guaranteed. Neither the public nor the sponsoring pharmaceutical firms have the financial means or the will to conduct such expensive clinical trails on numerous off-patent NSAIDs. Hence, our best alternative is to use these drugs only when they are needed and to monitor their adverse effects carefully. For example the use of selective COX-2 inhibitors for acute indications or when a simple analgesic such as acetaminophen would suffice is difficult to justify.
References
Bombardier C, Laine L, Reicin A, Shapiro D, Burgos-Vargas R, Davis B, Day R, Ferraz MB, Hawkey CJ, Hochberg MC, Kvien TK, Schnitzer TJ. Comparison of upper gastrointestinal toxicity of rofecoxib and naproxen in patients with rheumatoid arthritis. VIGOR Study Group. N Engl J Med 343:1520-1528, 2000.
Reicin AS, Shapiro D, Sperling RS. Comparison of cardiovascular thrombotic events in patients with osteoarthritis treated with rofecoxib versus nonselective nonsteroidal anti-inflammatory drugs (ibuprofen, diclofenac, and nabumetone). Am J Cardiol 89: 204-209, 2002.
Konstam MA, Weir MR, Reicin A, Shapiro D, Sperling RS, Barr E, Gertz BJ. Cardiovascular thrombotic events in controlled, clinical trials of rofecoxib. Circulation 104:2280-8, 2001.
Weir MR, Sperling RS, Reicin A, Gertz BJ. Selective COX-2 inhibition and cardiovascular effects: a review of the rofecoxib development program. Am Heart J 146:591-604, 2003.
http://www.dirc-canada.org/nddn/DIRC%20Care%20Advisory%20Vioxx.pdf
Vane JR, Bakhle YS, Botting RM. Cyclooxygenases 1 and 2. Annu Rev Pharmacol Toxicol 38:97-120, 1998.
Hensley, Scott: Pfizer to Test Celebrex as Heart Aid. http://www.wsj.com
Khan KN, Paulson SK, Verburg KM, Lefkowith JB, Maziasz TJ. Pharmacology of Cyclooxygenase-2 inhibition in the kidney. Kidney Int 61:1210-1219,2002.
Mayo PR, Skeith K, Russell AS, Jamali F Decreased dromotropic response to verapamil despite pronounced increased drug concentration in rheumatoid arthritis. Br J Clin Pharmacol 50:605-613, 2000.
Silverstein FE, Faich G, Goldstein JL, Simon LS, Pincus T, Whelton A, Makuch R, Eisen G, Agrawal NM, Stenson WF, Burr AM, Zhao WW, Kent JD, Lefkowith JB, Verburg KM, Geis GS. Gastrointestinal toxicity with celecoxib vs. nonsteroidal anti-inflammatory drugs for osteoarthritis and rheumatoid arthritis. The CLASS study: a randomized controlled trial. Celecoxib Long-Term Arthritis Safety Study. J Am Med Assoc 284: 1247–1255, 2000.
Davies NM, McLachlan AJ, Day RO, Williams KM. Clinical pharmacokinetics and pharmacodynamics of celecoxib: a selective cyclo-oxygenase-2 inhibitor. Clin Pharmacokinet 38:225-242, 2000.
Davies NM, Teng XW, Skjodt NM. Pharmacokinetics of rofecoxib: a specific cyclo-oxygenase-2 inhibitor. Clin Pharmacokinet 42:545-556,2003.
Corresponding Author: Corresponding Authors: Fakhreddin Jamali, Faculty of Pharmacy & Pharmaceutical Sciences, University of Alberta, Edmonton, Alberta, Canada T6G 2N8. fjamali@ualberta.ca
Neal Davies, Washington State University, College of Pharmacy, Department of Pharmaceutical Sciences, PO Box 646534, Pullman, Washington, USA 99164-6534. ndavies@wsu.eduPublished by the Canadian Society for Pharmaceutical Sciences.
Copyright © 1998 by the Canadian Society for Pharmaceutical Sciences.
http://www.ualberta.ca/~csps