J Pharm Pharmaceut Sci (www.ualberta.ca/~csps) 7(2):235-240, 2004
Liposomal prilocaine: preparation, characterization, and in vivo evaluation.
Cíntia Maria Saia Cereda, Daniele Ribeiro De Araujo, Giovana Bruschini Brunetto, Eneida De Paula1
Departamento de Bioquímica, Instituto de Biologia, Universidade Estadual de Campinas, UNICAMP, Campinas, SP, BrazilReceived 15 January 2004, Revised 9 June 2004, Accepted 23 June 2004, Published 15 July 2004
PDF Version
Abstract
PURPOSE: This study reports the development and in vivo evaluation of a liposomal system for the local anesthetic, prilocaine. METHODS. Liposomal prilocaine was prepared with egg phosphatidylcholine, cholesterol and α-tocopherol (4:3:0.07 molar ratio). The size of the liposomes was measured by laser light scattering and the effect of prilocaine on membrane fluidity made use of electron spin resonance (ESR). The anesthetic effect of liposomal prilocaine was compared to that of plain prilocaine solution (with or without vasoconstrictor) in a rat infraorbital nerve blockade model. RESULTS. Laser light-scattering analysis showed one major vesicle population of liposomes with ca 400 nm (100%), without size changes after prilocaine incorporation. The ESR results showed a decrease in the orientation of the phospholipid molecules into the liposomes (ca 11%) in the presence of prilocaine, which characterized the prilocaine-liposome interaction. A prolongation of anesthetic effect was produced by liposomal prilocaine in comparison to plain prilocaine (without vasoconstrictor, p<0.001). However, no statistical differences were found after comparison between liposomal prilocaine and vasoconstrictor-containing prilocaine. CONCLUSIONS. We suggest that the encapsulation of prilocaine in liposomes facilitates the controlled release of prilocaine (increasing the time of duration of the sensory nervous blockade) and constitutes a good choice to replace vasoconstrictor-containing local anesthetic formulations.
Introduction
Prilocaine is an aminoamide local anesthetic widely used in dentistry, usually associated with a vasoconstrictor to increase the anesthetic effect duration by keeping it longer at the site of action (1). In some cases, the use of vasoconstrictor is either not recommended or contraindicated (2-4) and longer-acting local anesthetics such as bupivacaine (5) present toxic side effects. Thus, the development of new local anesthetic formulations - intended to prolong the anesthetic action without increasing the systemic toxicity - is a current pharmaceutical challenge.
Liposomes are vesicles in which an aqueous core is entirely enclosed by surrounding lipid bilayers. They have been shown to be interesting as drug-delivery systems for local anesthetics since they enhance the availability of compounds, reduce their systemic toxicity and increase their half-life in vivo (6-8). Due to their similarity to biological membranes - since the lipid bilayer contains natural phospholipids and cholesterol - liposomes theoretically do not present any risk of antigenicity (9).
Local anesthetics have been encapsulated into liposomes and assayed in both animal and human models. Liposomal bupivacaine was evaluated in animal (9-11) or human models (6; 12) and liposomal lidocaine was used as for the epidural anesthesia in animals (13) or topical application in humans (14; 15). An increase of anesthetic effect was observed in all these studies, after administration of liposomal formulations. The present study comprises the preparation of a new liposomal prilocaine formulation, the physical and chemical characterization of the formulation and the assessment of its anesthetic efficacy in vivo, in an animal model.
Materials and Methods
Materials and animal model
3% Prilocaine Hydrochloride (PLC PLAIN ), 3% prilocaine hydrochloride plus 0.03 IU/ml felypressin (PLCFELYPRESSIN) and Thiopental were obtained from Cristália - Produtos Químicos e Farmacêuticos Ltda (SP, Brazil). Egg phosphatidylcholine (EPC), cholesterol (Ch) and α -tocopherol ( α -TC) were purchased from Sigma Chemical Co. (MO, USA). HEPES/NaCl buffer was prepared with analytical grade reagents.
Male Wistar rats, 250-350g, were obtained from CEMIB - UNICAMP (Centro de Bioterismo - State University of Campinas - UNICAMP) and were given free access to water and food throughout the study. The experiment was approved by the Institutional Committee for Ethics in Animal Research of the State University of Campinas - UNICAMP (Protocols 558-1 and 559-1), which follows the recommendations of the Guide for the Care and Use of Laboratory Animals.
Liposomal prilocaine characterization
Particle size determination
The mean diameter and size distribution of LUV suspension were analyzed by laser light scattering, using the Malvern Autosizer 4700 equipment. The measurements were made from a 90° angle at room temperature (25°C), before and after PLC incorporation.
Encapsulation efficiency and partition coefficient determination
The PLC was added to 4 mM liposomal suspensions, up to a 2 mM final concentration. Encapsulation efficiency was determined by centrifugation of the liposomal suspensions (120.000 x g, 2 h, 10ºC). PLC concentration in the supernatant was spectrophotometric determined at 224nm (molar extinction coefficient, ε = 5.10 3 M -1 , cm -1 ). The remaining concentration of PLC in the supernatant was subtracted from the initial PLC concentration, to define the amount bound to the lipid phase. The partition coefficient (P) was calculated from these data, according to equation 1:
(1)
Where: m = membrane phase, w = aqueous phase, n = number of moles of PLC and V = volume.
Membrane organization
The order parameter (S) of the liposomal membrane was measured in the spectra of 5-doxyl stearic acid (5-SASL) incorporated into LUV up to 2% molar ratio, before and after PLC encapsulation. The Electron Paramagnetic Resonance spectra were obtained with a Varian spectrometer operating at 9 GHz. Flat cells for aqueous solutions were used and the experiments were conducted at room temperature (22°C). For 5-SASL, whose long molecular axis is roughly parallel to the bilayer normal (16), the order parameter can be experimentally obtained according to the equation 2:
(2)
Where A// and A^ are hyperfine splittings corresponding to the spin label long molecular axis, parallel and perpendicularly oriented, respectively, to the external magnetic field. A// and A^ was measured as half the separation between the outer and the inner extrema, respectively (17) (Figure 1).
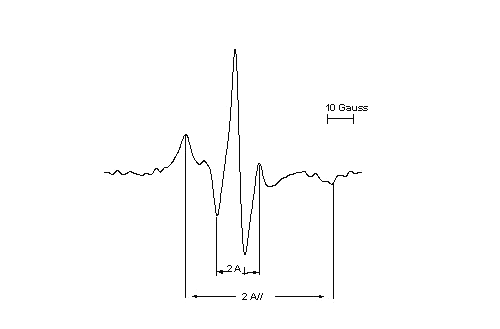
Figure 1: ESR spectrum of 5-doxyl stearic acid spin probe (2 mole %) into the egg phosphatidylcholine:cholesterol:a-tocopherol (4:3:0.07 mole %, 4 mM) liposomes, showing A// and A^ determination, according to (17).
A xx , A yy and A zz are the principal components of the hyperfine tensor and were taken as 6.0, 6.0 and 32.0 gauss, respectively (16).
In vitro release study
Prilocaine released from liposomes was investigated using a modified two-compartment in vitro method, described by Paavola et al (18). Briefly, the liposome formulation was introduced into a small donor compartment separated by a cellulose membrane (Spectra/Por 12 000 - 14 000 Da.) from a large acceptor compartment containing HEPES buffer, pH 7.4 (at 37°C). Aliquots (1 mL) were withdrawn from the acceptor compartment and the volume corrected for the withdrawn samples. PLC released was spectrophotometrically determined at 224 nm and expressed as percent values.
In vivo experiments
To evaluate anesthetic effect, the rat infraorbital nerve blockade technique adapted from Fink et al. (19) was used. The infraorbital nerve supplies upper lip and whisker area. The anesthetic effect was assessed by observation of aversive response to rat upper lip pinching according to the scores: 0 (aversive response) or 1 (no aversive response). These values were expressed as percent of local anesthetic activity. The infraorbital nerve emerges from the skull in the infraorbital notch, situated above a gap between the posterior four molars and the anterior incisor, in each side of the rat jaw. The tested anesthetic preparations were injected into this site after the animals were lightly anesthetized with intraperitoneal thiopental (25 mg/kg). Each group (n=7-10) received 0.1 mL of the following PLC preparations: Group I - prilocaine-free liposomes (LUVPRILOCAINE-FREE ); Group II - prilocaine solution (PLCPLAIN ); Group III - felypressin-containing prilocaine (PLCFELYPRESSIN ) and Group IV - liposomal prilocaine (PLCLUV ). The degree of sedation did not interfere with the generalized aversive response to the upper lip pinching with an artery forceps. Each test preparation was injected unilaterally into the right side. The intact left side served as control for comparing the responses from the two sides. All the experiments were performed by the same investigator. The animals were tested every 5-min up to the time when the first aversive sign in the injected side was detected. The efficacy of infraorbital nerve blockade was analyzed by time for sensory function recovery or duration of analgesia (time for recovery) and the total local anesthetic effect (estimated by the area under the effect vs. time curve, using the trapezoidal rule (20) and expressed by score/h - AUC), which were evaluated using the Origin 6.0 (Microcal TM Software, Inc.) program.
Statistical analysis
Size distribution of liposomes and in vitro release tests were analyzed by two-tailed unpaired t -test. Infraorbital nerve blockade data (AUC and time for recovery) were analyzed by the Kruskall-Wallis test and expressed as median (minimum and maximum limits). Statistical significance was defined as p < 0.05 (21).
Results and Discussion
Liposomal prilocaine characterization
Laser light scattering data revealed a single vesicle population, with 381.97±30.19nm (n=3), which is in accordance with the polycarbonate membrane pore used during the extrusion process. There were no size changes (406.07 ± 8.83 nm, n = 3) in the LUV, after PLC incorporation.
The average encapsulation rate of PLC into the liposomes and the partition coefficient (P) calculated from this data were 12.10 + 1.11% and 57 + 6% (mean + SD) at pH 7. 4, respectively.
These results represented the differences in the physicochemical properties of PLC, compared to other aminoamide anesthetics (22; 23). Prilocaine is one of the most hydrophilic local anesthetics presenting lower membrane partition values than other molecules such as mepivacaine, bupivacaine (24), benzocaine (25) and lidocaine (26). In fact, PLC incorporation is lower than that of bupivacaine (24.8 % + 4.2 %) (24) or lidocaine (19.1 % + 3.6 %) (Unpublished results) in the same LUV liposomes but its partition coefficient is in agreement with that determined in large multilamellar egg phosphatidylcholine vesicles (22). This amphiphilic character of PLC- also presented in other local anesthetics - makes it evident that a fraction of the anesthetic molecules resides in the lipid bilayer while the remainder is distributed in the aqueous phase.
The effect of PLC on the liposomal membrane organization was followed by ESR. PLC induced a decrease in the membrane order parameter ( ca 11%) (Figure 2) as also reported for other local anesthetic molecules (22; 27-29).
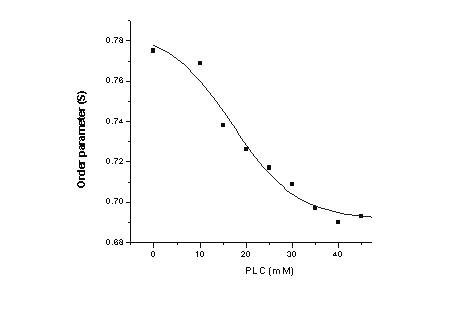
Figure 2: Effect of Prilocaine on the organization of the phospholipid molecules into the liposome bilayer (egg phosphatidylcholine:cholesterol:a tocopherol - 4:3:0.07 mole %, 4 mM). The order parameter was determined from the ESR spectra of 5-doxyl stearic acid incorporated into the liposomes (as described in methods).
The maximum effect was reached at a total PLC concentration of 40 mM that corresponds to 4.8 mM PLC inside the liposomes, according to the P value determined. At that point, a PLC-to-lipid molar ratio of 1.2:1 was reached in the bilayer, in agreement with the previous ESR results, leading to the saturation of the membrane phase, as discussed before (22).
In vitro drug release
Significant differences (p< 0.05) in the release kinetic patterns were observed between plain and liposomal PLC (Figure 3).
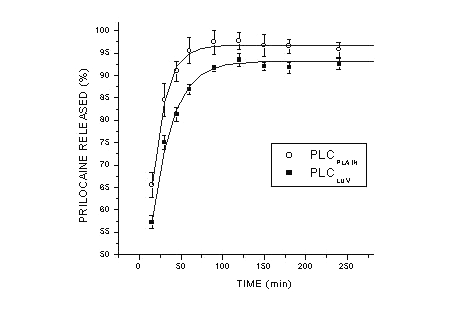
Figure 3: Release kinetic profiles (mean + SE) of plain and liposomal prilocaine formulations (n=4).
The liposomal formulation led to a slower release rate of PLC compared to its plain formulation. Equilibrium was delayed from 60 min (PLCPLAIN ) to 90 min with the drug-delivery system.
In vivo evaluation
Figure 4 shows that the duration of nerve blockade after PLCLUV treatment was significantly longer than the block produced by PLCPLAIN (p < 0.001) which is explained by longer residence-time of the liposomal formulation at the site of injection.
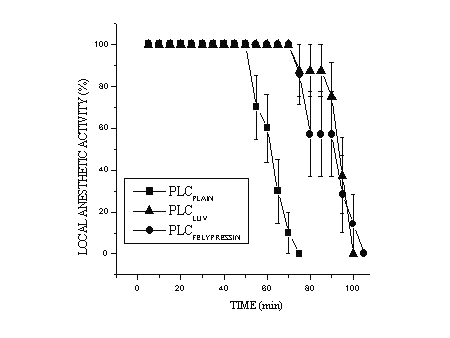
Figure 4: Local anesthetic activity - % (mean + SE) of plain, plus felypressin and liposomal prilocaine formulations, as evaluated with rat infraorbital nerve blockade.
Comparisons between PLCLUV and PLCFELYPRESSIN, did not show statistical differences. The total effect of nerve blockade (area under the curve - AUC) and recovery time for PLCPLAIN , PLCFELYPRESSIN and PLCLUV formulations are given in Table 1.
Table 1: Total effect of sensory blockade (AUC) and time for recovery for plain PLC (with and without vasoconstrictor) and PLCLUV formulations. Data are expressed as median (minimum - maximum) (n = 7 -10 /group).
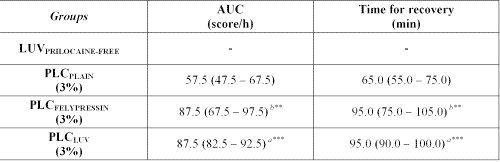
Statistical differences using a nonparametric analysis of variance (Kruskal-Wallis Test) between: a - PLCLUV vs. PLCPLAIN - p<0.001(***), b - PLCFelypressin vs. PLCPLAIN - p<0.01 (**). No significant differences were found between PLCLUV and PLCFelypressin .
The group that received LUVPRILOCAINE-FREE presented no effects while PLCPLAIN inducted nerve blockade up to 65 min after treatment. The nerve blockade induced by PLCLUV was clearly similar to that of PLCFELYPRESSIN and both prolong anesthesia up to 95 min. This result showed that liposomal formulation could be adopted in dentistry procedures, especially when the use of vasoconstrictors is contraindicated, such as in case of sulfite sensitivity, heart diseases, uncontrolled hyperthyroidism and diabetes, among other situations (2-4).
Conclusions
As shown by this study, the PLCLUV prolonged the duration of anesthesia when compared to the plain PLC (without vasoconstrictor). Besides, the anesthetic effect induced by the liposomal prilocaine formulation on an animal model was similar to that of vasoconstrictor-containing prilocaine. Thus, we suggest that liposomal PLC could replace vasoconstrictor-containing local anesthetics when the vasoactive compound is contraindicated. It is a new option for the controlled release of local anesthetics in dentistry, in the place of longer acting anesthetic such as bupivacaine, which induces toxicity to nervous and cardiovascular systems (5).
Acknowledgments
We would like to thank Dr Lúcia P. da Silva for her contributions reviewing this manuscript. C. M. S. Cereda was recipient of a fellowship from CAPES (Comissão de Aperfeiçoamento de Pessoal do Ensino Superior) and E. de Paula of a research fellowship from CNPq. This work was also supported by FAPESP (Proc. 96/04159-8, 01/12476-2).
References
Sisk, A. L., Vasoconstrictors in local anesthesia for dentistry. Anesth. Prog, 39:187-193, 1992.
Perusse, R.; Goulet, J.P.; Turcotte, J.Y., Contraindications to vasoconstrictors in dentistry: Part I. Cardiovascular diseases. Oral Surg. Oral Med. Oral Pathol.,74: 679-686, 1992.
Perusse, R.; Goulet, J.P.; Turcotte, J.Y. Contraindications to vasoconstrictors in dentistry: Part II. Hyperthyroidism, diabetes, sulfite sensitivity, cortico-dependent asthma, and pheochromocytoma. Oral Surg. Oral Med. Oral Pathol., 74: 687-691, 1992.
Goulet, J.P.; Perusse, R.; Turcotte, J.Y., Contraindications to vasoconstrictors in dentistry: Part III. Pharmacologic interactions. Oral Surg. Oral Med. Oral Pathol., 74: 692-697, 1992.
Mather, L.E.; Chang, D.H.T. Cardiotoxic with modern Local anesthetics. Is there a safer choice? Drugs, 61:333-342, 2001.
Boogaerts, J.G.; Lafont, N.D.; Declercq, A.G.; Hongwen, C.L.; Gravet, E.T.; Bianchi, J.A.; Legros F.J., Epidural administration of liposome-associated bupivacaine for the management of postsurgical pain: a first study. J. Clin. Anesth, 6: 315-320, 1994.
Grant, G.J., Bansinath, M., Liposomal delivery systems for Local Anesthetics. Reg. Anesth. Pain Med., 26: 61-63, 2001.
Grant, S.A., The Holy Grail: long-action local anesthetics and liposomes. Best Practice & Res. Clin. Anaesthesiology, 16: 345-352, 2002.
Malinovsky, J.M.; Benhamou, D.; Alafandy, M.; Mussini, J.M.; Coussaert, C.; Couarraze, G.; Pinaud, M.; Legros, F.J., Neurotoxicological assessment after intracisternal injection of liposomal bupivacaine in rabbits. Anesth. Analg., 85:1331-1336, 1997.
Boogaerts, J.G.; Lafont, N.D.; Luo, H.; Legros, F.J., Plasma concentrations of bupivacaine after brachial plexus administration of liposome-associated and plain solutions to rabbits. Can. J. Anaesth., 40: 1201-1204, 1993.
Grant, G.J.; Piskoun, B.; Lin, A.; Bansinath, M., An in vivo method for the quantitative evaluation of local anesthetics. J. Pharmac. Toxic. Methods, 43: 69-72, 2000.
Grant, G.J; Davidson, E.M.; Barenholz, Y. (2001) Lipossomal Bupivacaine produces prolonged analgesia in humans. Anesthesiology; 95:A926, 2001.
Mashimo T., Ushida I., Pack M., et al, Prolongation of canine epidural anesthesia by liposome encapsulation of lidocaine. Anesth. Analg., 74: 827-834, 1992.
Bucalo, B. D.; Mirikitani, E.J.; Moy, R. L., Comparison of skin anesthetic effect of liposomal lidocaine, nonliposomal lidocaine and EMLA using 30-minute application time. Dermatol. Surg., 24: 537-541 1998.
Finkel, J.C., Yang, C.I., Yarvitz, J. L., Patel, K.M., Neuroselective Sensory Electrodiagnostic Evaluation of 4% Liposomal Lidocaine. Anesth. Analg., 94: 1259-62, 2002.
Schreier, S.; Polnaszek, C. F.; Smith, I. C. P., Spin labels in membranes problems in practice. Biochim. Biophys. Acta, 515: 375-436, 1978.
Hubbell, W. L.; McConnell, H. M., Molecular motion in spin-labeled phospholipids and membranes. J. Am. Chem. Soc, 93: 314-320, 1971.
Paavola, A.; Yliruusi, J.; Kajimoto, Y.; Kalso, E.;Wahlström, T.; Rosenberg, P., Controlled release of lidocaine from injectables gels and efficacy in rat sciatic nerve block. Pharm. Res, 12: 1997-2002, 1995.
Fink, B.R., Aasheim, G., Kish, S.J., Croley, T.S., Neurokinetics of Lidocaine in Infraorbital Nerve of the Rat in Vivo. Anesthesiology, 42: 731-736, 1975.
Gantenbein, M.; Abat C.; Attolini, L.; Pisano, P.; Emperaire, N.; Bruguerolle, B. Ketamine effects on Bupivacaine local anaesthetic activity and pharmacokinetics of Bupivacaine in mice. Life Sciences, 61: 2027-2033, 1997.
Zar, J. H., Biostatistical Analysis , 3rd ed., New Jersey: Prentice Hall, 1996.
de Paula, E.; Schreier, S. Use of a novel method for determination of partition coefficient to compare the effect of local anesthetics on membrane structure. Biochim. Biophys. Acta, 1240: 25-33, 1995.
de Paula, E.; Schreier, S. Molecular and physicochemical aspects of local anesthetic-membrane interaction. Braz. J. Med. Res., 29: 877-894, 1996.
Araújo, D.R.; Pinto, L.M.A.; Cereda, C.M.S.; Brunetto, G. B.; Santana, M.H.A; de Paula, E. Encapsulation of mepivacaine prolongs the analgesia provided by sciatic nerve blockade in mice. Can. J. Anesth., 51: 566-572, 2004.
Pinto, L.M.A.; Yokaichiya, D.K.; Fraceto, L.F.; de Paula, E. Interaction of benzocaine with model membranes. Biophys. Chem., 87: 213-223, 2000.
Fraceto, L.F.; Pinto, L.M.A.; Franzoni, L.; Braga, A.A.C.; Spisni, A.; Schreier, S., de Paula, E. Spectroscopic evidence for a preferential location of lidocaine inside phospholipid bilayers. Biophys. Chem., 99: 229-243, 2002.
Frezzatti, Jr, W.A., Toselli, W.R. & Schreier, S., Spin label study of local anesthetic-membrane interactions. Phase separation of uncharged form and bilayer micellization by the charged form of tetracaine. Biochim. Biophys. Acta 860: 531-538, 1986.
Bianconi, M.L., Amaral, A.T. & Schreier, S.,Use of membrane spin label spectra to monitor rates of reaction of partitioning compounds. Hydrolysis of a local anesthetic analog. Biochem. Biophys. Res. Commun. 152: 344-350, 1988.
Lissi, E.; Bianconi, M.L.; Amaral, A.T.; de Paula, E.; Blanch, L.E.B.; Schreier, S. Methods for determination of partition coefficients based on the effect of solutes upon membrane structure. Biochim. Biophys. Acta, 1021: 46-50, 1990.
Corresponding Author: Eneida de Paula, Departamento de Bioquímica, Instituto de Biologia, UNICAMP, C. P. 6109, CEP 13083-970, Campinas, SP, Brazil. depaula@unicamp.br
Published by the Canadian Society for Pharmaceutical Sciences.
Copyright © 1998 by the Canadian Society for Pharmaceutical Sciences.
http://www.ualberta.ca/~csps
