J Pharm Pharmaceut Sci (www.cspscanada.org) 10(3):263-270, 2007
PREPARATION AND EVAL-UATION OF 211AT LABELLED ANTINEOPLASTIC ANTIBODIES
Almut Walte1, Siva Sankar Reddy Sriyapureddy1, Zekiye Korkmaz1, Doris Krull1, Oliver Bolte2, Michael Hofmann3, Geerd-J. Meyer1, Wolfram H. Knapp1
1 Klinik für Nuklearmedizin, Medizinische Hochschule Hannover, Hannover, Germany; 2 Klinik für Hämatologie, Hämostaseologie und Onkologie, Medizinische Hochschule Hannover, Hannover, Germany; 3 Klinik F Nuklearmedizin, Inselspital Bern, Suisse.
Received, January 1, 2007; Revised, April 8, 2007; Accepted, April 20 2007, Published April 22, 2007.
Corresponding Author: Geerd-J. Meyer, Klinik für Nuklearmedizin, Medizinische Hochschule Hannover, Carl-Neuberg-Str. 1, 30625 Hannover, Germany, Email: Meyer.Geerd-J@mh-hannover.de
ABSTRACT - PURPOSE. The objective of this study was to determine and verify the stability of 211At-labelled antibodies under physiological conditions and their specific cell-binding capacity for selected epitopes, in order to evaluate the potential of 211At for α-radioimmunotherapy. METHODS. 211At was produced at the departments cyclotron and was linked via the intermediate 3-211At-succinimidyl-benzoate (SAB) to the antineoplastic antibodies rituximab, gemtuzumab and gemtuzumab ozogamicin. The stability of the labelled antibodies was determined in serum for 21 h. Cell-binding experiments on HL-60 and CI-1 cells included kinetic, saturation and competitive binding studies. For comparison the binding to antigen-negative cells was determined. The binding specifity and affinity and the IC50-values were evaluated. RESULTS. A consistent yield of 30% and a specific activity of 3 MBq/nmol was obtained. The stability of 211At-antibodies in murine serum exceeded 85% at 37° C. Cell-binding to antigen-positive cells was >25%, while binding to antigen-negative cells did not exceed the unspecific binding and was smaller than 1%. IC50 values ranged between 2 and 11 nmol/L. CONCLUSIONS. A routine preparation of 211At-labelled antibodies was established and the stability of the 211At-labelled antibodies under physiologic conditions was verified. Apparently, labelling of antibodies with 211At by the method described does not compromise the affinity and specificity to the respective epitopes.
Introduction
Radioimmunotherapy combines the advantages of immunotherapy and radiation therapy. It has the high cytotoxic potential of radiation and can be used for treatment of systemic neoplasia. Therefore this concept is of particular interest for haematologic oncology. The selectivity of treatment is dependent on the specificity of the antibody as well as on the LET of the radionuclide.
Alpha-emitters are promising agents for radionuclide immunotherapy because of the high LET. Because of the halflife of 7.2 hrs complying with antibody biodistribution processes, 211At is of particular interest. The alpha particles from the decay of 211At have an average energy of 6.8 MeV and mean ranges in tissue of 55-80 µm. Their LETmean is 100 keV µm-1, which is associated with a high probability for creating strand breaks (1). In addition the great ionisation density along each track of an alpha-particle induces so many radicals, that the repair mechanisms of cells are overstrained. This phenomen is called by-stander-effect and leads to a higher rate of apoptosis than can be explained with direct hits of alpha-particles.
Relevant targets of haematological neoplasias are CD20 and CD33, present in lymphoma and leukaemia cells, respectively.
Rituximab (144 000 g/mol) is an antibody against the CD20 antigen which is located on B-lymphocytes, but not on haematopoietic stem cells or normal plasma cells. The antigen is expressed on >95% of all non-Hodgkin-lymphoma cells of type B. After binding to the antibody, the complex remains on the surface of the cell (2). Since 1998 rituximab is the treatment of lymphoma in Germany.
Gemtuzumab is a CD33 antibody with the molecular weight of about 150 000 Da. The CD33 antigen is expressed on the surface of leukaemic blasts in more than 80% of patients with acute myeloid leukaemia (AML). CD33 is also expressed on normal and leukaemic myeloid colony-forming cells, including leukaemic clonogenic precursors, but it is not expressed on pluripotent haematopoietic stem cells or on nonhaematopoietic cells, thereby limiting the haematological toxicity (3). Pure gemtuzumab is not a potent agent to eliminate leukaemia cells, but linked with the antibiotic ozogamicin, like in MylotargÒ, it is a licensed cytostatic drug. The CD33 antigen results in the formation of a complex that is internalized. GO is cytotoxic to the CD33 positive HL-60 human leukaemia cell line. GO produces significant inhibition of colony formation in cultures of adult leukaemic bone marrow cells. The cytotoxic effect on normal myeloid precursors leads to substantial myelosuppression, but this is reversible because pluripotent haematopoietic stem cells are spared (4).
The objective of this study was to determine and verify the stability of 211At-labelled antibodies under physiological conditions and their specific cell-binding capacity for the above epitopes, in order to evaluate the potential of 211At for α-radioimmunotherapy.
METHODS
Cell lines
The cell lines were purchased from DSMZ GmbH (Braunschweig, Germany). HL-60 cells were grown in RPMI-1640 medium containing 10% FCS , 100 IU/ml penicillin and 100 µg/ml streptomycin (Invitrogen, Karlsruhe, Germany). CI-1 cells were grown in RPMI-1640 medium containing 20% FCS, 100 IU/ml penicillin and 100 µg/ml streptomycin. All cells were maintained in an humidified atmosphere of 5% CO2/air at 37° C. Cell vitality was proved by staining with tryptane-blue.
Antibodies
Rituximab was obtained as MabThera® from Roche (Hertfordshire, UK) and stored at 4° C. Gemtuzumab was obtained and cleansed from BSA from Serotec (Kidlington, Oxford, UK) and stored in phosphate buffered solution (PBS, pH 7.4) at -20° C. GO was purchased as MylotargÒ from Wyeth Pharma GmbH (Münster, Germany) and stored at -20° C in ionic-free water.
Radionuclide and labelling
211At was produced at the departments cyclotron (MC35, Scanditronix) following the 209Bi(a,2n)211. At reaction, and dry distilled from the target matrix. In a first step it was reacted with N-succinimidyl 3-trimethylstannylbenzoate to give 3-211At-succinimidyl-benzoate (SAB). In a second step 0.1 to 1 mg of the particular antibody was labelled with the purified SAB. The labelled antibody was purified by flash-chromatography. The yield of radiolabelling was determined by radio-TLC (5,6).
Stability
The stability of the radiolabelled antibodies 211At-rituximab, 211At-gemtuzumab and 211At-labelled human IgG was determined in murine serum for three halflifes (21 h) at 37° C by gel-electrophoreses. The stability of the 211At-antibodies in serum was compared with that in NaCl-solution. The migration of intact antibodies were compared with that of fragments obtained by reduction.
Cell-binding experiments
For every cell experiment either a CD20-positive and CD33-negative human B-cell lymphoma CI-1 cell line or a CD33-positive and CD20-negative myeloid leukaemia HL-60 cell line were used. HL-60 cells are very sensitive to GO (7).
The kinetics of cell-binding were determined over a period up to 180 minutes. The specificity of cell-binding was tested in saturation and competitive binding experiments. All measurements were performed with three replicates and were carried out repeatedly.
On the day of the test, cells were seeded out in Eppendorf tubes, each with 1x106 cells in 200 µL PBS containing 2% FCS. Cells were put on ice until adding 200 µL of the active antibody (0.03 to 9.29 kBq) for incubation. The incubation time was 30 or 60 minutes under agitation at 37° C. For comparison anti-CD20-negative HL-60 cells were likewise treated with 211At-CD33-negative CI-1 cells were treated with 211At-GO and 211At-gemtuzumab, respectively. At the end of incubation cells were centrifuged, washed with ice-cold PBS and resuspended in 400 µL PBS. 200 µL of the cell suspension were taken off for measuring.
In saturation binding experiments the equilibrium binding was measured at various concentrations of the radioligand.
Unspecific binding of the 211At-antibodies to cells was obtained by measuring the radioligand binding at various concentrations of the radiolabelled antibody in the presence of an excess of the unlabelled antibody. Unlabelled antibody was added in a concentration equal to a 100 times the highest concentration of the radioligand to block virtually all specific binding. The rate of unspecific binding was calculated at various amounts of free radioligand. Specific radioligand binding was calculated by subtraction of the unspecific binding at a particular concentration of free radioligand from the total binding at that concentration.
In competitive binding experiments the binding of a single concentration of radioligand was measured at various concentrations of the unlabelled antibody. The unlabelled antibody was added in concentration steps ranging from 0.2 to 10 µg per 400 µL (3 to 167 nmol/L). The IC50 values of the labelled antibodies were determined by Eadie-Hofstee plots.
Activity measurement
The yield of chemical radiolabelling was measured in an ionization-chamber (Atomlab100plus dose calibrator, Biodex Medical Systems, Shirley, NY).
Activity bound to cells was measured for 5 minutes in a multi-crystal gamma counter (LB2111, EG&G Berthold, Wildbad, Germany) using the 125I- energy-channel. Cross calibration revealed an efficiency of the scintillation-counting of 20%. Background correction, cross-talk, and differences in the degree of efficiency of the detectors were carried out regularly.
Results
Chemical labelling yield
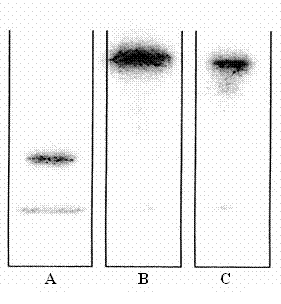
Figure 1: Gel-electrophoresis of 211At-rituximab: A: reduced 211At-rituximab, B: 211At-rituximab after 21 h at 37°C in murine serum, C: 211At-rituximab in 0.9% NaCl.
The labelling yield of rituximab after purification activity of 211At. The labelling yield of gemtuzumab and GO was 25% ±10%. The specific activity of the products ranged from 10 to 30 MBq/mg (1.5 to 4.5 MBq/nmol).
Stability in murine serum
The stability of all 211At-labelled antibodies over 21 h at 37° C in murine serum as determined by gel-electrophoreses was higher than 85%. The results and in particular the comparison of the intact antibodies with their fragments showed, that neither the antibodies were fragmented, nor astatine was removed and/or linked to other serum-proteins (Figure 1).
Cell-binding experiments
Maximum binding was found after 30 minutes and reached more than 10% for all antibodies with mean half maximum binding at 6 minutes.
The amount of bound activity depended on the specific activity of the labelled antibody, the amount of receptors on the cell-surface and the affinity of the antibody to these receptors. By adding increasing concentrations of the labelled antibody, the absolutely bound activity rose up to saturation and then levelled off. In contrast percentage binding decreased by increasing the quantity of added 211At-antibody (data not shown).
Unspecific binding was found to be proportional to the concentration of free radioligand. The corresponding factor for each radiolabelled antibody was determined by linear regression.
211At-Rituximab
Cell-binding formed a plateau after 30 to 60 minutes with half maximum binding at 5.5 ±1.5 minutes.
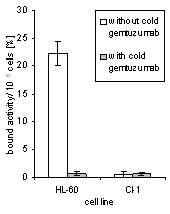
CI-1 cell-bound activity reached 31% when 2.6 kBq per 350 ng antibody were applied, whereas CD20-negative HL-60 cells only bound 0.25% of the added activity (Figure 2a). On average activity bound to CI-1 cells was more than 20fold higher when compared with CD20-negative cells. HL-60 cell-bound activity almost equalled the activity unspecifically bound to CI-1 cells.
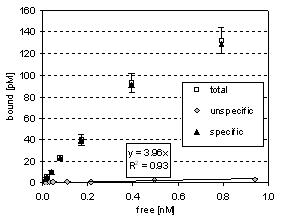
Maximal activity bound to CI-1 cells was reached with about 500 ng 211At-rituximab; in Figure 2b this value equals 4.2 kBq. The maximal cell-bound amount of rituximab exceeded 100 ng per 106 cells. Unspecific bound activity (determined by adding more than a 100fold excess of cold antibody) was linear with respect to the free radioligand concentration in solution (0.4%).
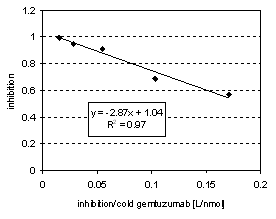
Competitive binding experiments revealed an IC50 value of 11 nmol/L by adding cold rituximab in concentration steps from 0 to 167 nmol/L to a constant concentration of 5.5 nmol/L 211At-rituximab (Figure 2c).
|
|
|
211At-GO
Cell-binding reached a plateau after 30 ±10 minutes with half maximum binding at 5 minutes.
The maximal bound mass was 10 ng 211At -GO per 106 cells which was reached by adding 60 ng 211At-GO.
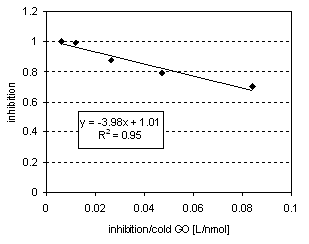
HL-60 cell-bound activity reached 26%, when 0.6 kBq per 30 ng antibody were applied. Percentage binding to CD33-negative CI-1 cells averaged only 0.7% (Figure 3a). Unspecific binding was found to be proportional to the concentration of unbound radioligand. The corresponding factor is 0.004 (Figure 3b). IC50 value was 4 nmol/L, determined by adding cold GO in concentration steps from 0 to 167 nmol/L, each with 2 nmol/L 211At-GO (Figure 3c).
|
211At-Gemtuzumab
Cell-binding reached a plateau after 30 ±10 minutes, with a half maximum at 6 minutes.
The maximal bound amount of 211At-gemtuzumab was similar to that of 211At-GO (20 ng per 106 cells by addition of 130 ng 211At-gemtuzumab).
Maximum HL-60 cell-bound activity reached 28% when 0.03 kBq per 2 ng antibody were applied (Figure 4a). Unspecific binding was a linear 0.004-fold fraction of the concentration of free ligand (Figure 4b).
IC50 value was 3 nmol/L, determined by adding cold gemtuzumab in concentration steps from 0 to 67 nmol/L, each with 0.2 nmol/L 211At-gemtuzumab (Figure 4c).
|
Figure 3c: Eadie-Hofstee Plot. Linear regression of the inhibition curve reveals an IC50 of 4 nmol/L with a regression coefficient of 95%. 1.2 kBq per 0.1 µg 211At-GO were applied to 106 cells. |
|
|
|
Discussion
Critical issues in the development of clinically effective α-radioimmunotherapy are the preservation of the binding properties and the verification of the in vivo stability of the labelled antibody under physiologic conditions. Radioimmunotherapy with 90Y and 131I has recently been introduced into clinical settings. Apparently the binding properties of the respective antibodies are not compromised by labelling with the above radionuclides.
Though 211At is extremely promising as a therapeutic radionuclide by its high LET a-emission, its use for protein labelling is challenging. Astatine has a very high atomic weight associated with a large radius which might cause conformational alterations of the molecule by labelling, and an electronic status that tends to form multiple hybridisation states, none of which exhibits strong covalent binding potentials.
Despite these obstacles 211At antibodies have been synthesized by using benzoic acid linkers and have been preclinically evaluated. Though the results were promising for further developments as a therapeutic tool, much more data need to be available, before 211At-labelled antibodies can be introduced in therapeutic regimens of haematological neoplasias. In particular no data are available concerning the fate of the labelled antibody under physiological conditions within a period of several halflifes. In addition, binding properties have to be evaluated with respect to individual radiochemical production settings and to the respective antibodies.
Using the two-step labelling method with SAB as described by Zalutsky (6) and Brechbiel (5) reproducible yields and a sufficient specific activity were obtained.
For three antibodies labelled with 211At we verified that more than 85% remained intact after incubation in murine serum at 37° C for three of the radionuclide. We have excluded the presence of significant amounts of decomposition products or free 211At.
All 211At-labelled antibodies showed a high specificity to the respective antigen-positive cells. Unspecific binding did not exceed 3% of the total binding under the condition that the specific binding sites were not completely occupied. The binding to antigen-negative cells did not exceed the unspecific binding. High affinity of all labelled antibodies has been verified by IC50 values in the range of a few nmol/L.
Because of the lower CD33 binding sites on HL-60 cells (about 10 000), when compared with the number of CD20 binding sites on CI-1 cells (more than 100 000), smaller amounts of 211At-gemtuzumab or 211At-GO were sufficient to achieve a high percentual binding. Accordingly the absolute amount of bound 211At-antibody was smaller in the case of the 211At-antiCD33-antibodies when compared to the 211At-antiCD20-antibody.
In order to achieve cytotoxicity by the radiolabelled antibody a sufficient number of alpha-particles have to traverse the target. (1.1 to 1.6 per cell). The average specific activity in our experiments was 3 MBq/nmol. This implies that only one out of 6000 antibodies is labelled with 211At (3 MBq correspond to 1011 atoms of 211At and 1 nmol antibody corresponds to 6x1014 molecules). Given that all binding sites are occupied, HL-60 cells are attached with 1.7 211At-atoms and CI-1 cells with 16.7 211At-atoms, respectively. Therefore the 211At density on CI-1 cells obtained in our settings is definitely in the cytotoxic range, whereas the 211At density on CI-1 cells is in the borderline range.
Conclusion
We have demonstrated the stability of three different 211At-labelled antibodies relevant for haematologic oncology under physiologic conditions. Furthermore all antibodies preserved their affinity and binding specificity to their antigen positive cells. The results of this study justify further preclinical evaluation of these 211At-labelled antibodies for α-radioimmuntherapy in animal experiments.
Acknowledgement
These studies were kindly supported by Deutsche José Carreras Leukämie-Stiftung e.V.
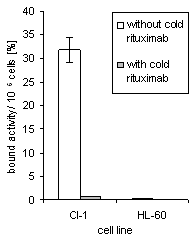
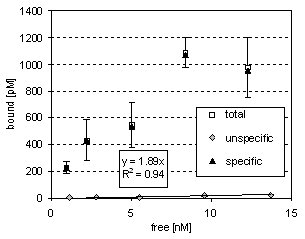 Figure 2b: Cell-bound 211At -rituximab related to the unbound activity in solution at equilibrium. Specific radioactivity was 8,4 MBq/mg. Linear Regression of the unspecific binding data (determined by adding cold rituximab) showed a slope of 0.0019 with a regression coefficient of 0.94.
Figure 2b: Cell-bound 211At -rituximab related to the unbound activity in solution at equilibrium. Specific radioactivity was 8,4 MBq/mg. Linear Regression of the unspecific binding data (determined by adding cold rituximab) showed a slope of 0.0019 with a regression coefficient of 0.94.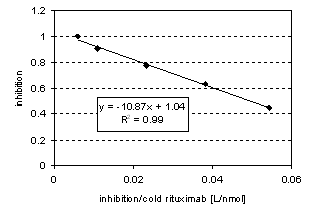 Figure 2c: Eadie-Hofstee Plot. Linear regression of the inhibition curve reveals an IC50 of 11 nmol/L with a regression coefficient of 99%. 7 kBq per 0.3 µg 211At-rituximab were applied to 106 cells.
Figure 2c: Eadie-Hofstee Plot. Linear regression of the inhibition curve reveals an IC50 of 11 nmol/L with a regression coefficient of 99%. 7 kBq per 0.3 µg 211At-rituximab were applied to 106 cells. 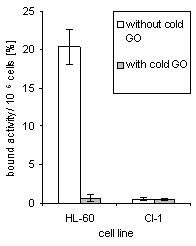
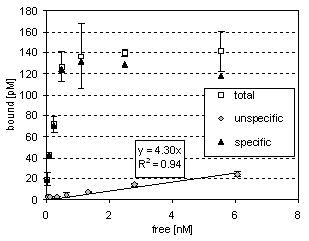 Figure 3b: Cell-bound 211At -GO related to the unbound activity in solution at equilibrium. Specific radioactivity was 9.9 MBq/mg. Linear regression of the unspecific binding data (determined by adding cold GO) showed a slope of 0.004 with a regression coefficient of 94%.
Figure 3b: Cell-bound 211At -GO related to the unbound activity in solution at equilibrium. Specific radioactivity was 9.9 MBq/mg. Linear regression of the unspecific binding data (determined by adding cold GO) showed a slope of 0.004 with a regression coefficient of 94%.